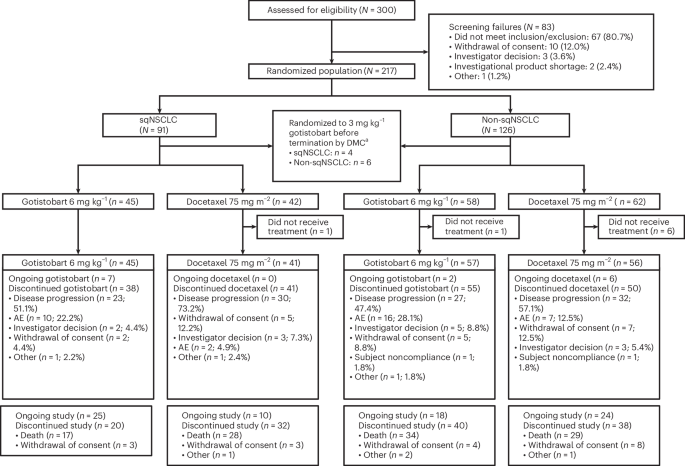
For patients battling advanced squamous non-minor cell lung cancer whose disease has become resistant to initial treatments, a new approach using the investigational drug gotistobart is showing promising signs of extending survival. Early results from the PRESERVE-003 trial, specifically its first stage, indicate that gotistobart may offer a benefit over standard chemotherapy with docetaxel. This is particularly significant as treatment options are limited for those whose cancer progresses after immunotherapy and chemotherapy.
Squamous cell carcinoma is a common type of lung cancer, accounting for roughly 30% of all cases, according to the American Cancer Society . When this cancer spreads – becomes metastatic – and stops responding to initial treatments, the prognosis is often poor. The PRESERVE-003 trial focuses on patients without specific genetic mutations that would make them eligible for targeted therapies, leaving them with few alternatives.
The initial stage of the trial involved patients who had already experienced disease progression after both immunotherapy and chemotherapy. Researchers found that those treated with gotistobart demonstrated encouraging overall survival outcomes compared to those receiving docetaxel. Although these findings are considered “nonpivotal” – meaning they aren’t definitive proof of the drug’s effectiveness – they provide a strong rationale for continuing to evaluate gotistobart in the ongoing phase 3 trial. Gotistobart is a next-generation anti-CTLA-4 agent, designed to work by enhancing the body’s immune response against cancer cells. What sets it apart is its pH-sensitivity, potentially allowing it to be more effective within the tumor microenvironment.
Understanding Gotistobart’s Mechanism
Current immunotherapies, which aim to unleash the immune system to fight cancer, don’t work for everyone. One reason is that cancer cells can create a suppressive environment that hinders immune cell activity. CTLA-4 is a protein that acts as a brake on the immune system. Anti-CTLA-4 antibodies, like gotistobart, aim to release that brake, allowing immune cells to attack cancer. However, traditional anti-CTLA-4 antibodies can cause significant side effects because they broadly affect the immune system throughout the body.
Gotistobart’s pH-sensitive design is intended to address this issue. Tumors often have a slightly acidic environment compared to normal tissues. The drug is designed to be more active in these acidic conditions, concentrating its immune-stimulating effects within the tumor itself and potentially reducing systemic side effects. This targeted approach could be a key advantage, offering a more effective and tolerable treatment option for patients with metastatic squamous non-small cell lung cancer.
PRESERVE-003 Trial: A Closer Look
The PRESERVE-003 trial is a randomized, phase 3 study designed to rigorously evaluate the efficacy and safety of gotistobart. The trial is being conducted in multiple centers, enrolling patients with advanced squamous non-small cell lung cancer who have progressed after prior immunotherapy and chemotherapy and do not have actionable genomic alterations. The first stage, as reported, focused on assessing initial outcomes and feasibility. The full trial will continue to accrue patients and follow them over time to determine if gotistobart can definitively improve overall survival and quality of life.
Researchers are carefully monitoring patients for side effects. While the initial data suggests a manageable safety profile, a larger and longer-term evaluation is needed to fully understand the potential risks and benefits of gotistobart. The trial’s design includes robust data collection on both efficacy and safety endpoints, providing a comprehensive assessment of the drug’s potential.
What So for Patients
The findings from stage 1 of the PRESERVE-003 trial offer a glimmer of hope for patients with limited treatment options. Metastatic squamous non-small cell lung cancer is a challenging disease, and new therapies are urgently needed. While gotistobart is still under investigation, the early results suggest it could represent a significant advance in the field. The concept of pH-sensitive immunotherapies is also gaining traction, with researchers exploring similar approaches for other types of cancer.
It’s important to remember that these are preliminary findings. The full results of the PRESERVE-003 trial are still awaited, and regulatory approval of gotistobart is not yet guaranteed. Patients interested in learning more about the trial or exploring potential treatment options should discuss their individual circumstances with their oncologist. Clinical trials, like PRESERVE-003, offer access to cutting-edge therapies and contribute to the advancement of cancer care.
Next Steps and Ongoing Research
The PRESERVE-003 trial is continuing to enroll patients, and the data safety monitoring board will regularly review the results. The next major checkpoint will be the analysis of the full trial dataset, which is expected to provide a definitive answer on whether gotistobart improves overall survival compared to docetaxel. Researchers are also exploring potential biomarkers that could help identify patients who are most likely to benefit from gotistobart, paving the way for more personalized treatment strategies. Further research is also underway to investigate the potential of combining gotistobart with other therapies, such as chemotherapy or other immunotherapies.
The development of new treatments for lung cancer is a complex and ongoing process. The PRESERVE-003 trial represents a significant step forward, but continued research and innovation are essential to improve outcomes for patients with this devastating disease. The hope is that gotistobart, or similar pH-sensitive immunotherapies, will ultimately become a valuable tool in the fight against lung cancer.
Disclaimer: This article is for informational purposes only and should not be considered medical advice. Please consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
Have you or a loved one been affected by lung cancer? Share your thoughts and experiences in the comments below. And please, share this article with anyone who might find it helpful.