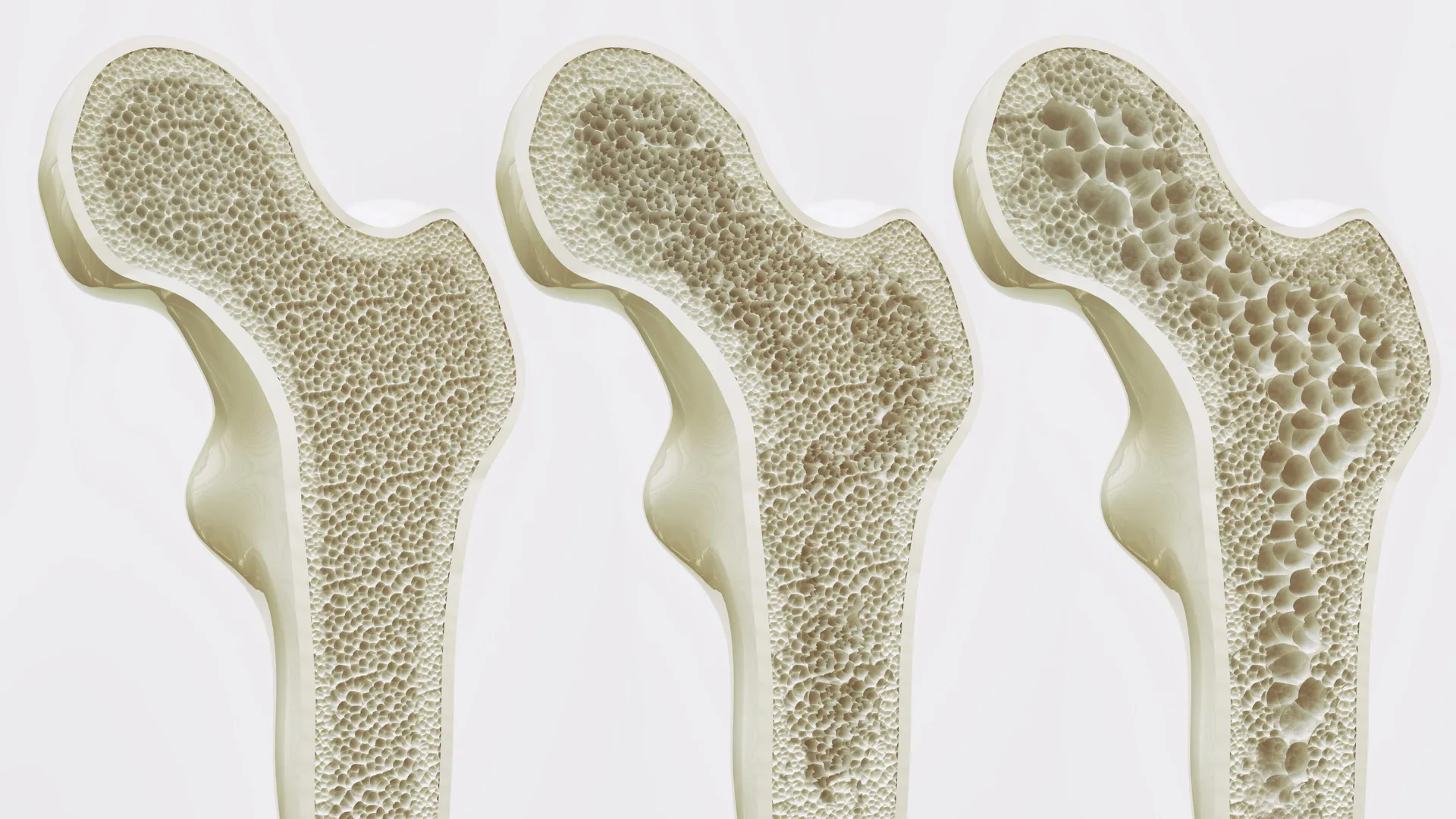
For many, the decline of bone health is a silent process, often unnoticed until a sudden fracture transforms a routine movement into a life-altering injury. This is the reality of osteoporosis, a condition that weakens the skeletal architecture and increases fragility, affecting millions worldwide. In Germany alone, approximately six million people live with the disease, with a disproportionate impact on post-menopausal women whose bone density naturally declines as estrogen levels drop.
Though, a breakthrough from researchers at Leipzig University suggests we may be moving toward a future where we can effectively maintain your bones strong for life. Rather than simply slowing the rate of bone loss, a newly identified biological target offers the possibility of actively rebuilding bone and restoring strength to those already suffering from skeletal degradation.
The discovery centers on a specific receptor known as GPR133. Part of a broader family called adhesion G protein-coupled receptors (GPCRs), these proteins act as cellular “antennae,” sitting on the surface of cells to receive and transmit signals that regulate vital bodily functions. While GPCRs are a cornerstone of modern pharmacology, this specific subgroup has remained largely mysterious until now.
By isolating the role of GPR133, scientists have identified a critical switch that controls how the body maintains its skeletal integrity. When this receptor is functioning correctly, it promotes the growth of dense, resilient bone; when it is impaired, the results mirror the onset of osteoporosis.
The Biological Tug-of-War: Building vs. Breaking
To understand why GPR133 is so significant, it is first necessary to understand that bone is not a static material, but a living tissue in a state of constant renewal. This process is managed by a delicate balance between two primary types of cells:

- Osteoblasts: The “builders” responsible for synthesizing new bone matrix.
- Osteoclasts: The “recyclers” that break down and absorb aged or damaged bone tissue.
In a healthy adult, these two forces exist in equilibrium. However, as we age or undergo hormonal shifts, the osteoclasts often begin to outpace the osteoblasts, leading to porous, brittle bones. The research from the Rudolf Schönheimer Institute of Biochemistry reveals that GPR133 is a key regulator of this balance.
The receptor is activated not only by chemical interactions between neighboring cells but too by physical stimuli, such as the pressure and movement associated with exercise. Once triggered, GPR133 sends signals that simultaneously ramp up the activity of bone-building osteoblasts and dampen the activity of bone-breaking osteoclasts. The result is a net gain in bone density and structural strength.
From Computer Screens to Bone Regeneration
The translation of this biological discovery into a potential therapy came through the use of advanced computer-assisted screening. Researchers identified a compound called AP503, a molecule designed to act as a stimulator for the GPR133 receptor, essentially mimicking the body’s natural bone-strengthening signals.
The efficacy of this approach was tested in animal models. Mice bred with genetic mutations that disrupted the GPR133 receptor developed low bone density early in life, closely mimicking human osteoporosis. When treated with AP503, these mice showed a significant increase in bone strength. Notably, the compound also worked in healthy mice, suggesting it could potentially be used preventatively to maintain skeletal density before loss begins.
“If this receptor is impaired by genetic changes, mice show signs of loss of bone density at an early age — similar to osteoporosis in humans. Using the substance AP503… We were able to significantly increase bone strength in both healthy and osteoporotic mice,” explains Professor Ines Liebscher, lead investigator of the study from the Rudolf Schönheimer Institute of Biochemistry at the Faculty of Medicine.
A Dual Benefit for the Aging Body
One of the most promising aspects of the GPR133 pathway is that its benefits may not be limited to the skeleton. For older adults, bone health is inextricably linked to muscle health; the loss of muscle mass (sarcopenia) often accompanies bone loss, creating a dangerous cycle that increases the risk of falls and subsequent fractures.
Earlier research by the Leipzig team found that activating GPR133 with AP503 also improved skeletal muscle strength. This dual-action potential—strengthening both the “frame” and the “engine” of the body—could be a game-changer for geriatric care, potentially preserving independence for aging populations by reducing the likelihood of mobility-related accidents.
This work is the culmination of over a decade of research conducted under the Collaborative Research Centre 1423, focusing on the structural dynamics of GPCR activation. By positioning Leipzig as a leader in this specialized field, the team has moved the conversation from managing osteoporosis to the possibility of reversing it.
Comparison of Bone Cell Activity
| Cell Type | Normal Function | Effect of GPR133 Activation | Resulting Bone State |
|---|---|---|---|
| Osteoblasts | Builds new bone | Increased Activity | Higher Density |
| Osteoclasts | Breaks down bone | Decreased Activity | Reduced Fragility |
The Path to Human Treatment
Despite the promising results in animal studies, the road to a clinical pharmacy shelf is long. The research team is currently conducting deeper investigations into how AP503 interacts with other systems in the body to ensure there are no adverse side effects. Because GPCRs are found in various tissues, the challenge lies in ensuring the treatment is targeted effectively.
For those currently managing bone loss, the identification of GPR133 provides a new roadmap. Current treatments often focus on inhibiting bone resorption (slowing the breakdown), but the AP503 approach focuses on anabolic stimulation (actively building). This shift in strategy could eventually lead to therapies that not only protect healthy bones but restore the skeletal integrity of those already affected by the disease.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
The research team at Leipzig University is continuing to refine the compound and explore the receptor’s function throughout the body. The next phase of research will focus on further detailing the molecular signaling of GPR133 to determine the precise dosage and delivery methods required for safety and efficacy before moving toward human trials.
Do you or a loved one manage bone health? Share your thoughts or questions in the comments below.