The landscape of American drug manufacturing is facing a potential shift as the federal government moves to prioritize domestic production. In a novel budget blueprint for 2027, the Trump administration has outlined a series of FDA-led policies designed to reduce reliance on foreign drug supplies and incentivize the development of pharmaceuticals within U.S. Borders.
These proposals target two critical stages of the drug lifecycle: the transition from laboratory research to human trials and the market entry of generic alternatives. By streamlining the path to clinical testing and introducing new protections for domestic generic makers, the administration aims to create a more resilient national supply chain.
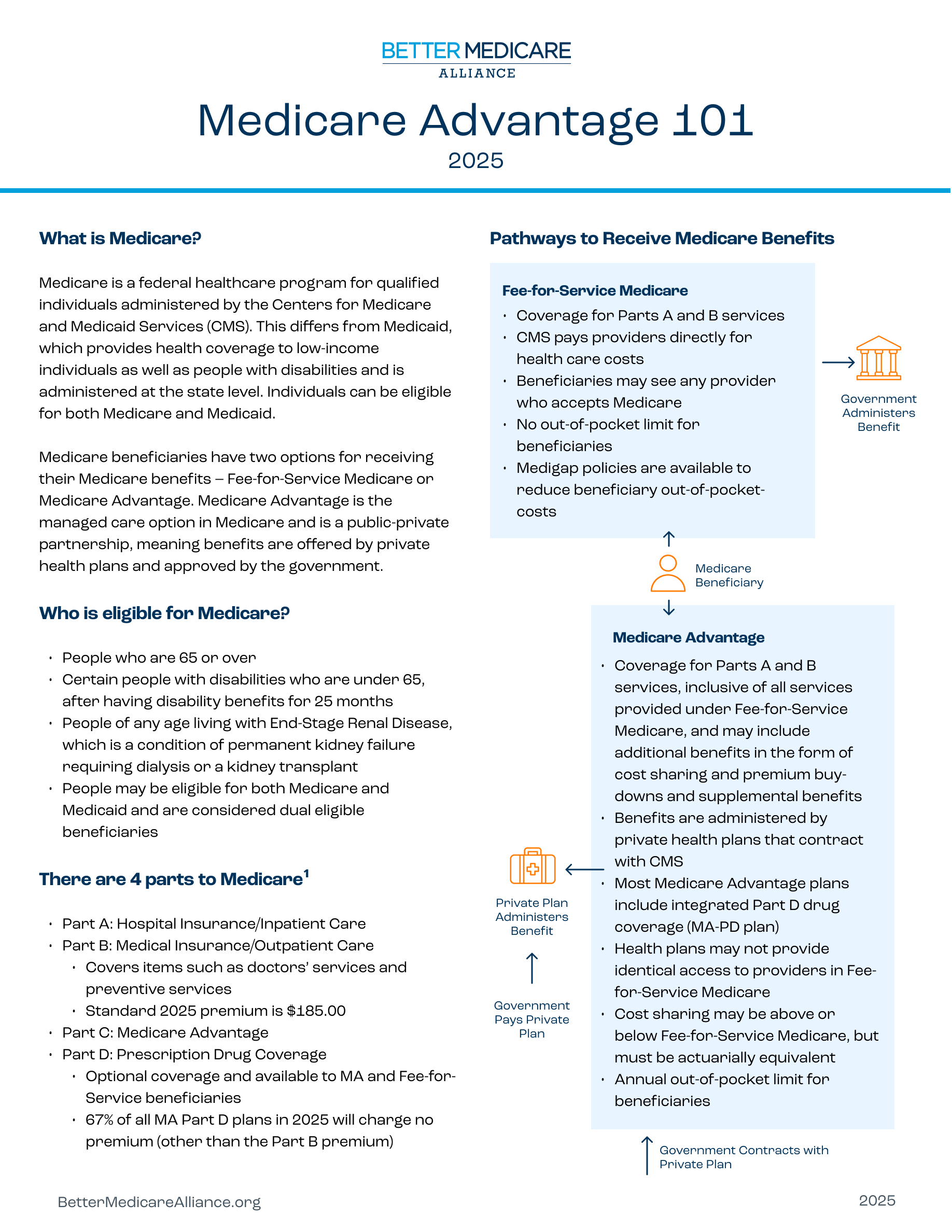
Even as the budget blueprint focuses on broad infrastructure and policy shifts, the pharmaceutical industry remains preoccupied with the accessibility of high-demand treatments. For millions of patients, the intersection of federal policy and private insurance—specifically Wegovy and Medicare Advantage coverage—remains the most pressing concern in the current healthcare climate.
As a physician, I have seen how the gap between FDA approval and actual patient access can create significant clinical hurdles. When a drug like Wegovy (semaglutide) is approved for chronic weight management, the medical benefit is clear, but the financial architecture of Medicare Advantage plans often determines who actually receives the medication.
Proposed FDA Shifts to Boost Domestic Manufacturing
The core of the 2027 budget blueprint is a strategic push to bring the “making” of medicine back to the United States. The FDA is proposing to simplify the regulatory hurdles that drugmakers face when moving into clinical testing on U.S. Soil. Currently, many companies conduct early-phase trials abroad where costs may be lower or regulatory environments differ, but the administration argues that domestic testing accelerates the eventual rollout of these therapies to American patients.
Perhaps more significant for the broader market is the proposal to grant an “exclusivity” period to U.S.-based generics manufacturers. In the pharmaceutical world, exclusivity is a powerful tool; it prevents other competitors from entering the market for a set period, allowing the original manufacturer to recoup investment costs. By extending this benefit specifically to domestic firms, the government hopes to develop U.S.-based manufacturing financially competitive with overseas hubs, particularly in Asia.
The goal is to mitigate the risks of drug shortages, which have plagued the U.S. Healthcare system in recent years, ranging from chemotherapy agents to basic antibiotics. A domestic supply chain is viewed not just as an economic priority, but as a matter of national security.
The Access Gap: Wegovy and Medicare Advantage
While the administration focuses on how drugs are made, patients are focused on how they are paid for. The struggle to secure coverage for Wegovy through Medicare Advantage plans highlights a systemic tension in U.S. Healthcare. Unlike traditional Medicare, which historically has not covered weight-loss medications, Medicare Advantage plans—offered by private insurers—have more flexibility in what they cover.
Still, this flexibility has led to a fragmented landscape. Some Medicare Advantage plans provide robust coverage for GLP-1 agonists, while others maintain strict exclusions or require rigorous “step therapy,” forcing patients to strive cheaper, less effective medications before qualifying for Wegovy. This creates a disparity in care where a patient’s health outcome is tied directly to their specific insurance provider rather than clinical need.
For clinicians, this means that the “prescription” is only the first step. The second, and often more difficult step, is navigating the prior authorization process. The high cost of these medications puts pressure on private insurers, who are tasked with balancing patient health outcomes against the rising cost of pharmacy benefits.
Understanding the Impact on Stakeholders
The shift toward domestic manufacturing and the battle over drug coverage affect different groups in distinct ways:
- Patients: May see more stable drug availability if domestic manufacturing increases, but continue to face “coverage lotteries” depending on their Medicare Advantage plan.
- Drug Manufacturers: Could benefit from faster clinical trial transitions and exclusivity windows, potentially offsetting the higher cost of U.S. Labor and facilities.
- Generic Makers: U.S.-based firms stand to gain a competitive edge over international rivals if the proposed exclusivity periods are implemented.
- Healthcare Providers: Will likely continue to manage the administrative burden of securing approvals for high-cost medications like Wegovy.
Comparing Policy Goals and Patient Realities
The following table outlines the contrast between the administration’s high-level manufacturing goals and the immediate challenges faced by patients accessing modern biotech.
| Focus Area | Administrative Goal (2027 Blueprint) | Patient/Clinical Reality |
|---|---|---|
| Manufacturing | Increase U.S.-based drug production | Frequent shortages of critical meds |
| Clinical Trials | Easier transition to U.S. Testing | Long wait times for new drug access |
| Generics | Exclusivity for domestic makers | High cost of branded GLP-1s |
| Coverage | Broad economic resilience | Variable Medicare Advantage access |
What This Means for the Future of Biotech
The convergence of these policies suggests a move toward a “closed-loop” pharmaceutical ecosystem where the U.S. Controls the research, the testing, and the manufacturing. If the FDA successfully implements the 2027 budget proposals, the U.S. Could see a surge in domestic biotech infrastructure. However, manufacturing capacity does not automatically equal affordability.
The real test will be whether the increase in domestic production leads to lower costs for the consumer. If the government provides exclusivity to domestic generics, it may temporarily limit competition to reward U.S. Firms—a move that could potentially keep prices higher in the short term even as the supply chain becomes more secure.
For those tracking Wegovy and Medicare Advantage, the focus remains on the Centers for Medicare & Medicaid Services (CMS) and how they will regulate the “supplemental benefits” that private plans offer. Any shift in federal guidelines regarding obesity as a chronic disease could fundamentally change the coverage mandates for all Medicare Advantage plans.
Disclaimer: This article is for informational purposes only and does not constitute medical or financial advice. Please consult a licensed healthcare provider or insurance specialist for guidance on specific medications and coverage.
The next critical checkpoint will be the formal review of the 2027 budget blueprint by Congress, where the specific terms of the “exclusivity” periods for generic manufacturers will be debated and potentially modified.
We want to hear from you. Have you experienced challenges securing coverage for GLP-1 medications through your insurance? Share your thoughts in the comments below.

