When a patient is admitted with severe COVID-19, the clinical priority is often immediate viral suppression to prevent respiratory failure. In these high-stakes environments, Veklury (remdesivir) has remained a cornerstone of antiviral therapy. However, for the prescribing physician, the challenge is rarely the drug itself, but rather the complex chemical environment of a patient already taking multiple medications for comorbidities like hypertension, diabetes, or chronic kidney disease.
Understanding Veklury drug interactions is not merely a matter of checking a box; This proves a critical safety protocol. Because remdesivir is administered intravenously and metabolized primarily in the liver, its interaction profile can influence both the drug’s efficacy and the patient’s overall stability. For clinicians and pharmacists, the goal is to balance the urgency of treating a viral infection with the necessity of avoiding adverse drug-drug interactions (DDIs) that could lead to organ toxicity or therapeutic failure.
As a board-certified physician, I have observed that the most dangerous interactions are often those that are overlooked in the rush of an emergency admission. While remdesivir is generally well-tolerated, its interaction with certain antimalarials and its impact on renal and hepatic functions require a nuanced approach to patient management. Ensuring that a patient’s medication list is scrubbed for contraindications is the first and most vital step in the administration of this antiviral.
The Primary Conflict: Chloroquine and Hydroxychloroquine
The most significant and well-documented interaction involving Veklury is with chloroquine and hydroxychloroquine. Early in the pandemic, these drugs were explored as potential treatments, but subsequent research revealed a critical pharmacological conflict. These antimalarials can significantly reduce the antiviral activity of remdesivir, effectively neutralizing the benefit of the treatment.
The mechanism behind this interaction is rooted in the way these drugs affect the intracellular environment. Remdesivir requires specific cellular conditions to be converted from its prodrug form into the active triphosphate metabolite that inhibits the viral RNA polymerase. Chloroquine and hydroxychloroquine alter these conditions, hindering the activation of the drug. The FDA-approved labeling suggests avoiding the co-administration of these agents to ensure maximum therapeutic efficacy.
While the use of hydroxychloroquine for COVID-19 has largely fallen out of favor in clinical guidelines, it remains a standard treatment for autoimmune conditions such as rheumatoid arthritis and lupus. In these cases, clinicians must carefully weigh the necessity of the autoimmune medication against the necessitate for antiviral therapy, often opting to temporarily pause the antimalarial during the remdesivir course.
Managing Hepatic and Renal Vulnerabilities
Beyond direct drug-drug conflicts, the safety of Veklury is heavily dependent on the patient’s organ function. Remdesivir is associated with elevations in liver enzymes, specifically alanine aminotransferase (ALT). When a patient is concurrently taking other hepatotoxic drugs—such as certain statins, methotrexate, or high-dose acetaminophen—the risk of drug-induced liver injury (DILI) increases.
Clinical monitoring is essential. The NIH COVID-19 Treatment Guidelines emphasize the importance of baseline liver function tests (LFTs) before starting therapy. If ALT levels increase significantly (typically more than 10 times the upper limit of normal), the drug should be discontinued to prevent acute liver failure.
Renal function presents a different set of challenges. Remdesivir is formulated with an excipient called sulfobutyl ether beta-cyclodextrin (SBECD), which is cleared by the kidneys. In patients with severe renal impairment, SBECD can accumulate in the body. While the direct toxicity of SBECD is a subject of ongoing study, the potential for accumulation means that patients with a very low estimated glomerular filtration rate (eGFR) require extreme caution or may be ineligible for the drug.
Summary of Interaction Levels for Veklury
| Interaction Level | Example Agent | Clinical Impact | Management Action |
|---|---|---|---|
| Major | Hydroxychloroquine | Reduced antiviral efficacy | Avoid co-administration |
| Moderate | Hepatotoxic Drugs | Increased risk of liver injury | Frequent LFT monitoring |
| Moderate | Renal Impairment | SBECD accumulation | Monitor eGFR closely |
| Minor | CYP3A4 Inducers | Potential decrease in plasma levels | Clinical observation |
Clinical Monitoring and Best Practices
To mitigate the risks associated with Veklury drug interactions, a structured monitoring timeline is recommended. This approach ensures that any adverse reaction is caught before it becomes a critical event. The focus is typically divided between hepatic surveillance and renal tracking.
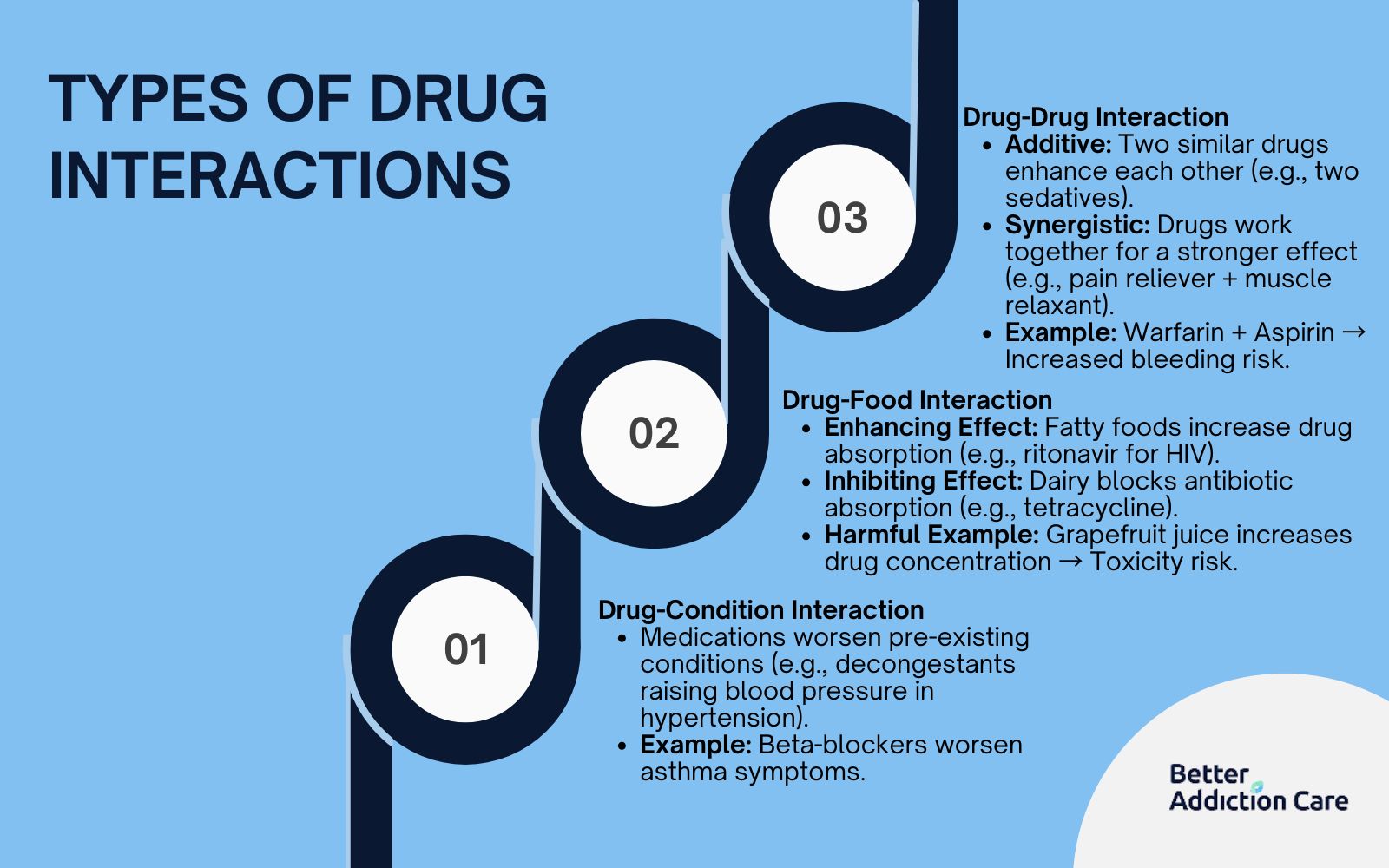
For hepatic safety, clinicians typically order LFTs on day 1 and day 7 of the treatment cycle. This allows the medical team to identify a trend of rising transaminases early. If a patient is on a regimen of other medications that stress the liver, this monitoring may be increased to every 48 to 72 hours.
For renal safety, the focus is on the eGFR. If a patient’s kidney function declines during the course of treatment, the risk of SBECD accumulation rises. Physicians must decide whether the benefit of continuing the antiviral outweighs the risk of excipient buildup, particularly in patients already receiving other nephrotoxic agents like aminoglycosides or certain contrast dyes.
Pharmacists play a pivotal role in this process. By performing a comprehensive medication reconciliation upon admission, they can identify “silent” interactions—such as over-the-counter supplements or home medications—that the patient may have forgotten to mention but which could interfere with the drug’s metabolism or exacerbate its side effects.
The Broader Impact on Patient Outcomes
The goal of managing these interactions is to ensure that the patient receives the full benefit of the antiviral without incurring avoidable harm. When remdesivir is used in isolation or with carefully managed co-medications, it has shown a capacity to shorten recovery time in hospitalized patients. However, when interactions are ignored, the resulting toxicity can lead to longer hospital stays and increased morbidity.
The complexity of treating COVID-19 in the elderly—who often present with “polypharmacy” (the use of five or more medications)—makes the study of these interactions even more vital. For these patients, the interaction isn’t just between two drugs, but between a drug, a disease state, and a diminished physiological reserve.
For healthcare providers seeking the most current prescribing information, the official manufacturer data from Gilead Sciences provides the most detailed breakdown of pharmacokinetic studies and updated safety warnings.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or medication.
As the landscape of COVID-19 evolves, the medical community continues to monitor the long-term effects of antiviral therapies and their interactions with emerging treatments. The next major checkpoint for clinical updates will be the upcoming revisions to the NIH treatment guidelines, which will likely incorporate new data on the use of remdesivir in combination with newer monoclonal antibodies and oral antivirals.
Do you have questions about medication safety or current antiviral protocols? Share your thoughts in the comments or share this guide with a colleague.