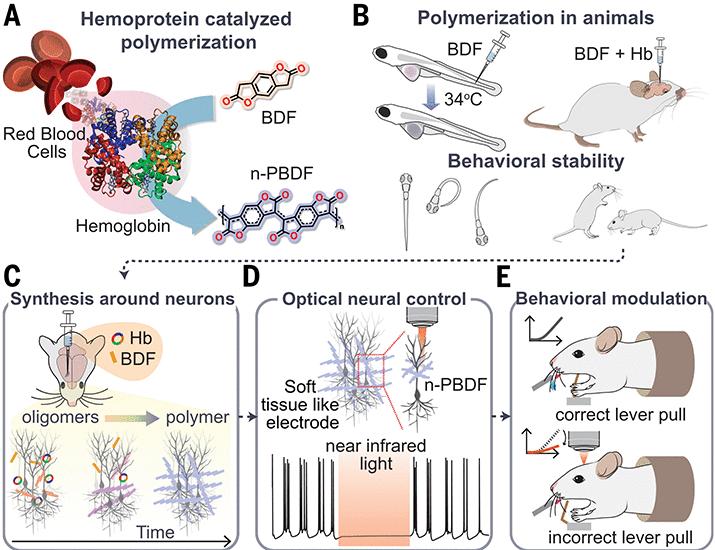
Bridging the gap between rigid synthetic electronics and the soft, gelatinous environment of the human brain has long been one of the most stubborn hurdles in bioelectronics. Traditional implants often trigger an immune response, leading to glial scarring that eventually insulates the device from the very neurons This proves meant to monitor or control. However, a novel approach utilizing blood-catalyzed n-doped polymers for neural control may offer a way to integrate electronics directly into living tissue without the need for invasive, rigid substrates.
Researchers have developed a method to assemble conducting polymer interfaces in vivo, using the body’s own blood as a catalyst to create a seamless electrical bridge. This breakthrough, detailed in the journal Science, allows for the reversible optical control of neurons, meaning scientists can use light to switch neural activity on and off with high precision and minimal tissue trauma.
For clinicians and researchers, the significance lies in the “substrate-free” nature of the interface. Rather than inserting a pre-fabricated needle or wire, the conducting polymer is assembled in place. This reduces the mechanical mismatch between the device and the brain, potentially eliminating the chronic inflammation that typically plagues long-term brain-machine interfaces.
Overcoming the ‘N-Doping’ Barrier
In the world of conducting polymers, “doping” is the process of adding impurities to a material to change its electrical properties. Most biocompatible conducting polymers are p-doped (positive charge carriers), but n-doped polymers (negative charge carriers) are notoriously difficult to stabilize in biological environments. What we have is because n-doped materials tend to react aggressively with oxygen and water, causing them to degrade almost instantly upon contact with living tissue.

The researchers bypassed this instability by using a blood-catalyzed assembly process. By leveraging specific chemical components within the blood to trigger the polymerization, they were able to create n-doped interfaces that remain stable and functional within the brain. This chemical stability is what enables the polymer to act as an efficient transducer, converting optical signals into the electrical impulses that neurons understand.
This capability represents a significant shift in how we approach neural modulation. Even as optogenetics—the use of light to control neurons—has been a gold standard in research, it typically requires genetic modification of the cells to make them light-sensitive. This blood-catalyzed polymer approach provides a synthetic alternative that could theoretically work in non-genetically modified cells, broadening the potential for clinical application.
Reversible Optical Control and Precision
The primary utility of these polymers is their ability to facilitate reversible optical neural control. In practical terms, So the interface can be used to either excite or inhibit neuronal firing depending on the light stimulus applied. This “bidirectional” control is critical for treating neurological disorders where the goal is not just to stimulate a dormant area of the brain, but to quiet an overactive one.
The mechanism works by utilizing the n-doped polymer as a light-to-electricity converter. When exposed to specific wavelengths of light, the polymer generates a local electrical field that alters the membrane potential of adjacent neurons. Because the polymer is integrated at the molecular level with the tissue, the current injection is highly localized, reducing “off-target” effects that often occur with traditional deep-brain stimulation electrodes.
Comparing Neural Interface Technologies
The shift from rigid electrodes to substrate-free polymers changes the fundamental interaction between the device and the central nervous system.
| Feature | Traditional Microelectrodes | Blood-Catalyzed Polymers |
|---|---|---|
| Mechanical Property | Rigid/Metallic | Soft/Organic |
| Tissue Response | High (Glial Scarring) | Low (Biocompatible) |
| Installation | Surgical Insertion | In vivo Assembly |
| Control Mechanism | Electrical Current | Optical-to-Electrical |
Clinical Implications and Future Constraints
The ability to create a seamless, n-doped interface has immediate implications for the treatment of refractory epilepsy, Parkinson’s disease, and spinal cord injuries. By providing a way to modulate neural circuits without the risk of chronic inflammation, this technology could lead to implants that last a lifetime rather than a few years.
However, several constraints remain before this can transition to human trials. The precision of the “in vivo assembly” must be perfected to ensure that the polymer forms exactly where intended without migrating to other areas of the brain. The long-term metabolic fate of these synthetic polymers—how the body eventually breaks them down or clears them—requires extensive longitudinal study to ensure Notice no delayed toxicological effects.
Researchers are now focusing on the stability of these interfaces over months and years. While the initial results show a marked decrease in the immune response, the ultimate test will be whether the n-doped state can be maintained indefinitely in the oxygen-rich environment of the cerebral cortex.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
The next critical milestone for this research will be the publication of long-term biocompatibility data in larger animal models, which will determine if the substrate-free approach can maintain its electrical integrity over extended periods. Updates on these trials are expected to be presented at upcoming neuroengineering symposiums.
Do you think synthetic interfaces are the future of brain-machine interaction? Share your thoughts in the comments or share this story with your network.