In the complex battle against cancer, the immune system relies on specialized “scout” cells to identify malignant threats and alert the rest of the body to attack. Among these, conventional type 1 dendritic cells (cDC1s) are considered essential for antitumor immunity, acting as the primary bridge between the tumor microenvironment and the T cells that execute the kill.
Yet, tumors are notoriously hostile environments. They create metabolic “dead zones” that can starve or disable immune cells, often rendering these critical cDC1s dysfunctional. New research published in Science reveals that the secret to a dendritic cell’s success in these conditions lies in its mitochondria—the cell’s energy powerhouses—and how their metabolic signaling directs dendritic cell function in antitumor immunity.
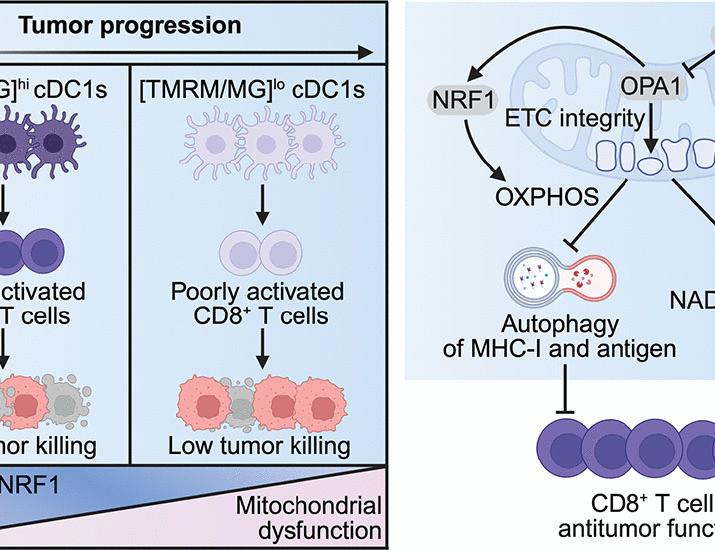
The study demonstrates that cDC1s within a tumor do not exist in a single state of health or dysfunction. Instead, they exhibit discrete mitochondrial states that determine whether the cell will effectively activate the immune system or succumb to the suppressive pressures of the cancer. By understanding these metabolic switches, researchers may be able to “flip” the switch toward a more aggressive immune response.
For clinicians and patients, this discovery moves the conversation beyond simply increasing the number of immune cells in a tumor. It suggests that the quality and metabolic fitness of those cells are the true determinants of whether a treatment, such as immunotherapy, will succeed or fail.
The Metabolic Tug-of-War Inside the Tumor
To understand why this discovery matters, one must first understand the “metabolic competition” occurring within a tumor. Cancer cells consume glucose and nutrients at an accelerated rate, leaving very little for the infiltrating immune cells. This nutrient deprivation often leads to mitochondrial stress, which can shut down the cDC1’s ability to present antigens—the molecular “fingerprints” of the cancer—to T cells.
The researchers found that intratumoral cDC1s are not a monolithic group. Some maintain high mitochondrial fitness, allowing them to sustain the energy-intensive process of antigen presentation and T-cell activation. Others enter a state of mitochondrial dysfunction, where the signaling pathways that normally trigger an immune response are silenced.
This metabolic signaling acts as a regulatory gate. When mitochondria are functioning optimally, they produce specific metabolites and signals that inform the dendritic cell to remain active and “pro-inflammatory.” When these mitochondria fail, the cell may not only stop working but could potentially contribute to an environment that helps the tumor hide from the immune system.
Decoding the Role of cDC1s in Cancer Immunity
Dendritic cells are the sentinels of the immune system. The cDC1 subtype is particularly vital due to the fact that it specializes in “cross-presentation,” a process where it takes up tumor antigens and presents them to CD8+ T cells, the primary “killer” cells of the immune system. Without functional cDC1s, the T cells remain blind to the tumor’s presence.
The study highlights a critical gap in previous knowledge: although we knew cDC1s were necessary, we didn’t know how they maintained their “functional fitness” while submerged in the toxic chemistry of a tumor. The identification of discrete mitochondrial states provides a map of how these cells survive and thrive, or fail, in the face of malignancy.
The implications for immunotherapy are significant. Many current treatments, such as checkpoint inhibitors, rely on the presence of already-activated T cells. However, if the cDC1s are metabolically exhausted, the “spark” required to ignite the T-cell response is missing. Targeting the mitochondrial metabolism of these cells could potentially prime the tumor for a much more effective response to existing drugs.
Comparing Mitochondrial States in cDC1s
| Mitochondrial State | Metabolic Profile | Immune Outcome |
|---|---|---|
| High Fitness | Efficient ATP production & signaling | Strong T-cell activation; tumor regression |
| Dysfunctional | Metabolic stress; reduced signaling | Immune evasion; tumor growth |
| Intermediate | Variable nutrient uptake | Unstable or transient immune response |
From Laboratory Discovery to Clinical Application
The transition from a molecular discovery in Science to a bedside treatment is often a long road, but the path for metabolic reprogramming is becoming clearer. By identifying the specific signaling molecules that maintain mitochondrial health in cDC1s, scientists can explore pharmacological ways to protect these cells from the tumor’s suppressive effects.

Potential strategies emerging from this line of research include:
- Metabolic Supplementation: Providing specific nutrients or precursors that support mitochondrial respiration in dendritic cells.
- Small Molecule Activators: Using drugs to mimic the “high fitness” signals that maintain cDC1s active.
- Combination Therapies: Pairing metabolic boosters with traditional immunotherapies to ensure the “scouts” (cDC1s) are ready to lead the “soldiers” (T cells).
This approach shifts the focus of oncology from simply attacking the tumor to supporting the host’s internal defense mechanisms. It recognizes that the immune system is not a static tool, but a dynamic biological entity that can be tuned and optimized through metabolic intervention.
As the medical community continues to investigate these pathways, the goal is to move toward a more personalized form of immunotherapy—one where a patient’s tumor is analyzed not just for its genetic mutations, but for its metabolic landscape, allowing doctors to tailor the “metabolic fuel” provided to the immune system.
Disclaimer: This article is provided for informational purposes only and does not constitute medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
The next phase of research will likely focus on validating these mitochondrial markers in human clinical trials to determine if targeting cDC1 metabolism can improve survival rates in patients with “cold” tumors—those that currently do not respond to immunotherapy. Further updates on these clinical milestones are expected as researchers move from murine models to human cohorts.
Do you have questions about the future of metabolic immunotherapy? Share this article and join the conversation in the comments below.