Researchers are uncovering a sophisticated biological strategy used by Vibrio cholerae, the bacterium responsible for cholera, to survive in the face of viral attacks. Novel evidence suggests that DNA uptake in cholera may increase defense mechanisms by allowing the bacteria to scavenge genetic material from their environment, effectively “updating” their immune systems to counter predatory viruses known as bacteriophages.
This process, known as natural transformation, allows the bacteria to integrate foreign DNA into their own genome. While bacteria frequently exchange genetic information, the specific use of this mechanism to acquire defenses against phages highlights an ongoing evolutionary struggle. By absorbing DNA from deceased neighbors or other related species, V. Cholerae can rapidly acquire new tools to block viral entry or degrade viral DNA once it has entered the cell.
As a physician and medical writer, I have observed that understanding these microbial “arms races” is critical for public health. When bacteria evolve more robust defense mechanisms, it can complicate the development of phage therapies—a promising alternative to antibiotics—and provide insights into how pandemic strains of cholera persist and spread globally.
The stakes are significant. Cholera remains a global threat, particularly in regions with inadequate water and sanitation. The ability of the pathogen to evolve its defenses in real-time means that the bacteria are not static targets; they are dynamic organisms capable of rapid adaptation to survive both the human immune system and the viruses that hunt them.
The Evolutionary Arms Race: Bacteria vs. Phages
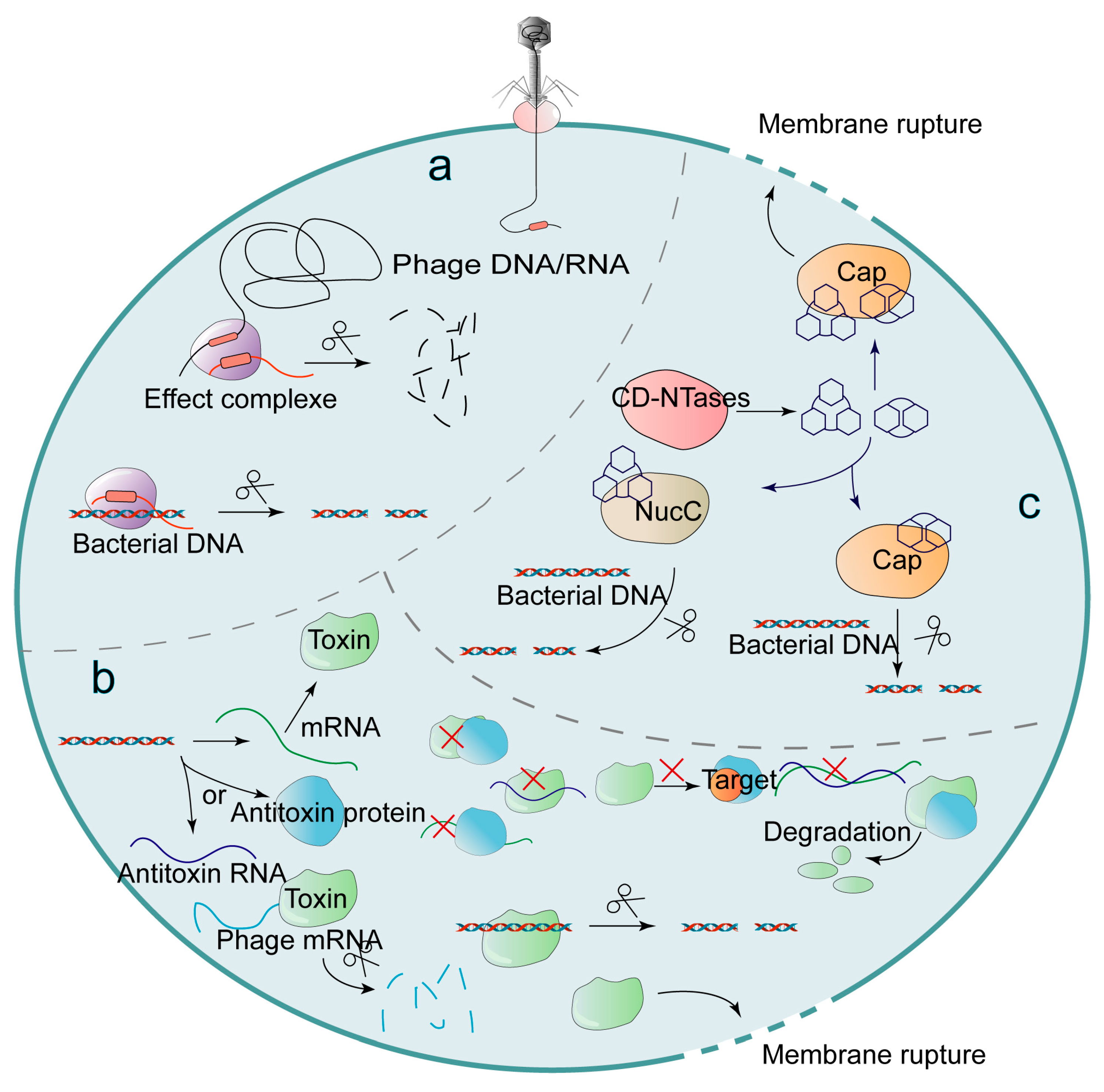
The relationship between Vibrio cholerae and bacteriophages is a classic biological conflict. Phages are viruses that infect bacteria, often killing them in the process. To survive, V. Cholerae has developed a variety of defense systems, including CRISPR-Cas systems, which act like a molecular “memory” of past infections, allowing the bacteria to recognize and chop up viral DNA upon second contact.
But, phages as well evolve, developing ways to bypass these defenses. This creates a continuous cycle of adaptation. The discovery that V. Cholerae can utilize DNA uptake to enhance its defenses adds a new layer to this conflict. Instead of relying solely on random mutations or the slow process of vertical inheritance, the bacteria can essentially “crowdsource” survival strategies from the surrounding microbial community.
This genetic exchange is not limited to the bacteria’s own species. Research indicates that V. Cholerae can swap defenses with its neighbors, integrating genetic sequences that provide resistance to specific phages. This horizontal gene transfer allows a population of bacteria to develop into resistant to a virus much faster than would be possible through individual mutation.
Mechanisms of Genetic Acquisition
The process of natural transformation involves several key steps that enable the bacteria to acquire and utilize new defensive traits:
- Competence: The bacteria enter a state of “competence,” where they express specific proteins that allow them to bind and pull DNA from the external environment into the cell.
- Integration: Once inside, the foreign DNA is integrated into the bacterial chromosome through homologous recombination.
- Expression: The new genes are transcribed and translated into proteins, such as surface receptors that prevent phages from attaching or enzymes that degrade viral genetic material.
This ability to incorporate external DNA is particularly potent in the nutrient-rich but volatile environments where cholera thrives, such as estuarine waters and the human gut. In these settings, the turnover of microbial life is high, providing a constant supply of “environmental DNA” for the bacteria to sample.
Global Impact and Pandemic Evolution
The implications of these defense mechanisms extend beyond the laboratory. The evolution of pandemic cholera is closely tied to how the bacteria adapt to their environment. By studying the genetic markers of these defenses, scientists can better track the movement and evolution of the pathogen at its global sources.
Research published in Nature has emphasized that the evolution of pandemic strains is often driven by the acquisition of new genetic elements. When V. Cholerae enhances its defense mechanisms through DNA uptake, it may not only survive viral predation but also become more resilient to other environmental stressors, potentially increasing its virulence or persistence in the environment.
| Mechanism | Source of Information | Primary Function |
|---|---|---|
| CRISPR-Cas | Internal Memory | Recognizes and cuts specific viral DNA sequences. |
| Natural Transformation | Environmental DNA | Acquires new resistance genes from other bacteria. |
| Surface Modification | Mutation/Acquisition | Changes “locks” on the cell wall to block phage entry. |
This adaptability is why cholera remains difficult to eradicate. The bacteria are essentially operating a biological software update system, constantly refining their defenses to stay one step ahead of the viruses that would otherwise keep their populations in check.
What This Means for Future Treatment
From a clinical perspective, the fact that DNA uptake in cholera may increase defense mechanisms is a double-edged sword. On one hand, it provides a roadmap for understanding how the bacteria function, which could lead to new ways to disable these defenses. It suggests that “phage therapy”—using viruses to kill antibiotic-resistant bacteria—will require a highly flexible approach.
If V. Cholerae can rapidly acquire resistance to a specific phage by absorbing DNA from its environment, a single-phage treatment may quickly become ineffective. This suggests that “phage cocktails”—mixtures of multiple different viruses—may be necessary to overwhelm the bacteria’s ability to adapt. By attacking the bacteria through multiple different pathways simultaneously, clinicians can make it harder for the pathogen to find a single genetic “fix” to survive.
understanding the triggers that make V. Cholerae “competent” for DNA uptake could allow researchers to develop inhibitors that prevent the bacteria from updating their defenses, making them more susceptible to both phages and traditional antimicrobial treatments.
Disclaimer: This article is provided for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
As genomic surveillance continues to improve, the next major milestone will be the real-time mapping of these genetic exchanges during active outbreaks. Public health agencies and researchers are currently working to refine the speed at which they can sequence environmental samples to identify emerging resistance patterns before they lead to widespread treatment failure.
We invite you to share your thoughts on these developments in the comments below and share this article with colleagues in the public health and biotechnology sectors.