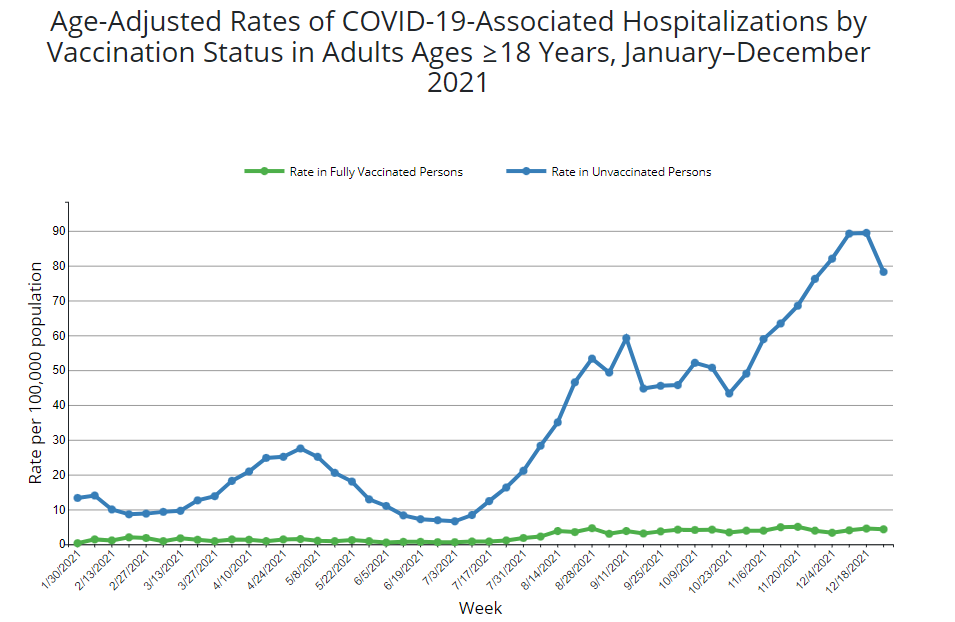
Public health transparency has come under renewed scrutiny following reports that a senior official at the Centers for Disease Control and Prevention (CDC) delayed the release of a critical research report. The document in question provided evidence that the updated Covid-19 vaccine significantly reduced the risk of hospitalizations and emergency room visits during the previous winter season.
The delay of this data creates a tension between the internal review processes of federal health agencies and the public’s need for timely, evidence-based information. For clinicians and patients, knowing the precise effectiveness of a booster shot is not merely an academic exercise. it is the foundation upon which vaccination decisions are made during peak respiratory virus seasons.
The report focused on the effectiveness of the 2023-2024 formula, specifically analyzing how the shots performed against circulating variants. According to the findings, those who received the updated vaccine were substantially less likely to experience severe outcomes compared to those who remained unvaccinated or had not received the most recent dose. Despite these clear results, the acting head of the agency held the report, preventing its publication in the Morbidity and Mortality Weekly Report (MMWR), the CDC’s primary vehicle for scientific communication.
The Friction Between Review and Release
In the world of public health, the gap between data collection and publication is often where political and administrative pressures collide. The acting head’s decision to stall the report has raised questions about whether the delay was a matter of rigorous scientific vetting or a strategic choice regarding the timing of the message.
From a medical perspective, the “effectiveness” of a vaccine is a moving target. As the virus mutates, the window of maximum utility for a booster is narrow. When data showing high effectiveness is withheld, public health officials lose a primary tool for combating vaccine hesitancy. The report’s findings—that the shots worked as intended to keep people out of hospitals—served as a powerful counter-narrative to claims that the vaccines had lost their utility.
The internal process at the CDC typically involves several layers of review to ensure that the data is robust and the conclusions are not overreached. However, the scale of the delay in this instance has been characterized by some as an unnecessary hurdle that obscured a clear public health win. The stakeholders affected by this delay include not only the general public but also healthcare providers who rely on MMWR data to guide their patient consultations.
Timeline of the Report’s Progression
While the exact dates of internal drafts are often shielded from public view, the trajectory of the report followed a specific sequence of administrative hurdles:
| Phase | Action/Status | Impact |
|---|---|---|
| Data Analysis | Research completed on winter hospitalizations. | Confirmed vaccine effectiveness. |
| Submission | Report submitted for internal agency review. | Ready for public dissemination. |
| Administrative Hold | Acting head delays publication. | Data remains internal; public unaware. |
| Eventual Release | Report finally published via MMWR. | Delayed evidence reaches the public. |
Analyzing the Impact on Vaccine Confidence
The delay of a report on Covid vaccine effectiveness occurs at a time when trust in institutional health guidance is fragile. When a government agency sits on positive data, it can inadvertently fuel theories that the data was being manipulated or that the results were less favorable than they actually were.
For a board-certified physician, the ability to say “the data shows a significant reduction in hospitalization” is the most effective way to encourage a patient to receive a booster. When that data is missing for months, the conversation shifts from evidence to anecdote. The report’s conclusion that the vaccine reduced emergency visits is a critical metric because it speaks directly to the burden on the healthcare system, not just individual health.
The acting head’s role involves balancing the agency’s reputation for accuracy with the urgency of the current health landscape. However, the risk of “over-polishing” a report can lead to a loss of relevance. In the case of respiratory viruses, a report published in the spring about a winter surge is far less impactful than one published in November.
What This Means for Public Health Policy
The implications of this delay extend beyond a single report. It highlights a systemic vulnerability in how the U.S. Government communicates scientific success. If the process for releasing positive data is as cumbersome as the process for releasing cautionary data, the public receives a skewed perception of the vaccine’s performance.
- Clinical Guidance: Doctors may have missed the opportunity to use the most current data to persuade high-risk patients.
- Public Trust: Perceptions of “gatekeeping” scientific data can erode confidence in the Centers for Disease Control and Prevention.
- Policy Planning: State and local health departments rely on these reports to allocate resources for the following season.
The core of the issue is the “information vacuum.” In the absence of official CDC data, the public often turns to unverified sources or fragmented data from other countries, which may not apply to the U.S. Population or the specific variants circulating domestically.
The Path Toward Greater Transparency
To prevent future delays, You’ll see calls for more streamlined “fast-track” publication processes for data that has a direct impact on immediate clinical decisions. By creating a clear distinction between long-term academic studies and urgent public health surveillance, the agency could ensure that life-saving information reaches the public in real-time.
The current situation underscores the need for a culture of transparency where data—whether it confirms a hypothesis or challenges it—is released promptly. The acting head’s decision to delay a report that showed the vaccine was working effectively suggests a caution that may have outweighed the benefit of public awareness.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Please consult a healthcare provider for personalized medical guidance regarding vaccinations.
The next expected development will be the CDC’s updated guidance for the upcoming autumn vaccination campaign, which will likely incorporate the delayed data from the previous winter to justify new booster recommendations. This official update will serve as a benchmark for whether the agency has adjusted its communication timeline.
We invite you to share this story and join the conversation in the comments about the balance between scientific review and public transparency.