For patients battling non-muscle-invasive bladder cancer, the period following surgery is often defined by a grueling cycle of surveillance and uncertainty. Despite successful early intervention, this specific form of the disease is notorious for its high recurrence rate, leaving patients in a state of perpetual anxiety, awaiting the next cystoscopy to see if the cancer has returned.
A new approach developed by researchers at Stanford University could fundamentally change this trajectory. By utilizing a sophisticated test urinaire (urine test) to detect circulating tumor DNA, clinicians may soon be able to identify “minimal residual disease”—microscopic traces of cancer that remain after treatment—long before a tumor becomes visible to the naked eye during a clinical exam.
The study, published in the journal Cell, suggests that a simple urine sample could serve as a molecular compass, guiding doctors toward a more personalized treatment plan. The goal is to distinguish between patients who are truly cured by surgery alone and those who require aggressive follow-up therapy, potentially sparing thousands from unnecessary and taxing medical procedures.
As a physician and medical writer, I have seen how the “one size fits all” approach to cancer surveillance can inadvertently harm patient quality of life. The ability to quantify the risk of relapse at a molecular level represents a shift from reactive medicine—treating the tumor once it reappears—to proactive management.
The Challenge of the ‘BCG Cycle’ and Bladder Cancer Recurrence
Most patients diagnosed with non-muscle-invasive bladder cancer (NMIBC) undergo a standard protocol: an endoscopic resection to remove the tumor, followed by a series of six weekly instillations of Bacillus Calmette-Guérin (BCG), a form of immunotherapy designed to stimulate the immune system to attack remaining cancer cells.
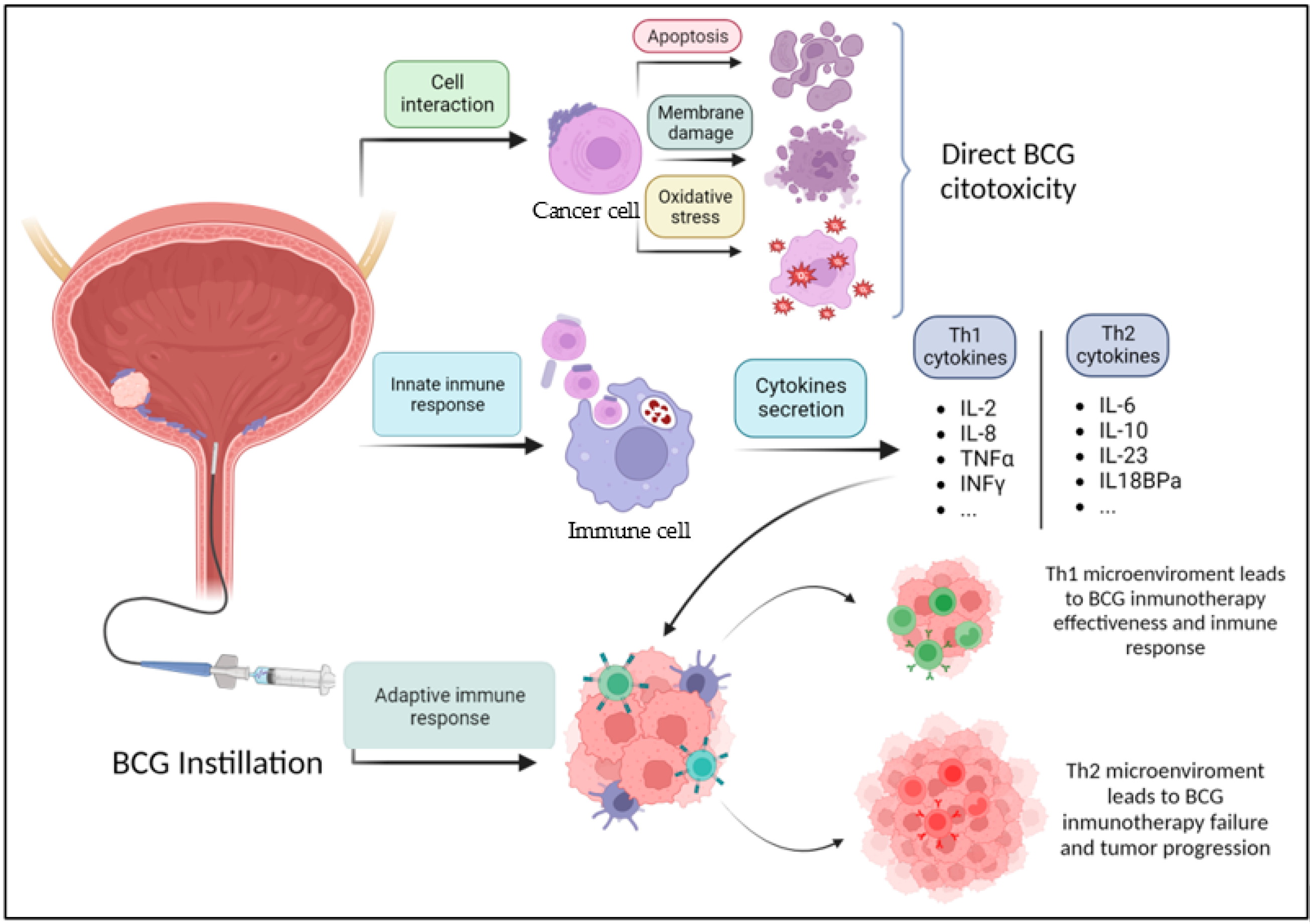
While BCG is effective for many, We see not without significant drawbacks. The treatment can cause severe bladder irritation and systemic side effects. The global healthcare system has faced recurring BCG shortages, making consistent follow-up difficult for many clinics. The central dilemma for urologists has been that they cannot currently determine which patients are already cured by surgery and could safely skip BCG, and which patients are at such high risk that they need intensified therapy.
Until now, the primary tool for monitoring has been the cystoscopy—a procedure where a camera is inserted into the bladder. While gold-standard, it only detects visible masses. It cannot “see” the molecular signatures of a relapse that is just beginning to form.
Overcoming ‘Clonal Cystopoiesis’: The Noise of Aging
The transition to “liquid biopsies” (detecting cancer via fluids like blood or urine) is not without technical hurdles. The Stanford team encountered a specific biological obstacle: clonal cystopoiesis. This phenomenon occurs when healthy individuals, particularly as they age, develop mutations in the lining of the bladder (the urothelium) that mimic the genetic markers of cancer, even in the absence of a tumor.
This “biological noise” has historically led to false positives in urinary DNA tests, making it difficult for researchers to tell the difference between a benign age-related mutation and a true sign of returning cancer.
To solve this, the research team implemented a specialized statistical method to filter out this background noise. By isolating the specific signal of the tumor from the general mutations associated with aging, they created a high-precision tool for detecting minimal residual disease.
“Our test allows for the non-invasive detection of minimal residual disease after bladder cancer treatment, while accounting for mutations present in the normal urothelium, which has complicated previous studies,” stated Dr. Joseph Liao, MD, professor of urology and co-author of the study. “For the first time, we were able to distinguish patients likely cured by BCG from those cured by surgery.”
Clinical Impact: From 261 Patients to Personalized Care
The researchers validated this method by analyzing 261 patients with NMIBC. Urine samples were collected at three critical stages: before surgery, immediately following surgery, and after the completion of BCG therapy. The results provided a clear correlation between the presence of tumor DNA and the likelihood of relapse.
Patients whose urine still contained tumor DNA after BCG treatment faced a significantly higher risk of recurrence. Conversely, those whose samples were clear of tumor DNA showed excellent clinical outcomes. Most importantly, the test identified risks of relapse in several cases where the standard cystoscopy appeared completely normal, proving that molecular detection can precede visual detection.
| Feature | Standard Surveillance (Cystoscopy) | Stanford Urine DNA Test |
|---|---|---|
| Detection Method | Visual identification of masses | Detection of circulating tumor DNA |
| Invasiveness | Invasive (Catheter/Camera) | Non-invasive (Urine sample) |
| Timing | Detects visible tumors | Detects minimal residual disease (MRD) |
| Clinical Goal | Confirm presence of tumor | Predict risk of future recurrence |
The Future of Precision Urology
The implications of this test urinaire extend beyond simple detection. If integrated into standard clinical practice, this technology could allow urologists to stratify patients into distinct risk categories. Those with a “molecularly clear” status after surgery might avoid the side effects and costs of BCG entirely. Meanwhile, high-risk patients could be transitioned to more aggressive therapies or enrolled in clinical trials for new medications before their cancer progresses to a more dangerous, muscle-invasive stage.
Dr. Eila C. Skinner, MD, professor of urology and chair of the Department of Urology at Stanford, emphasized the necessity of these biomarkers. She noted that as new, expensive, and potentially toxic treatments emerge, the ability to match the right therapy to the right patient is essential to avoid over-treatment and optimize survival rates.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Patients should consult their healthcare provider for diagnosis and treatment options regarding bladder cancer.
The next step for this research involves larger prospective trials to determine exactly how much the use of this test improves long-term survival rates and reduces the incidence of over-treatment. Further data will be required before this liquid biopsy becomes a routine part of the urological standard of care.
We invite you to share your thoughts or experiences with cancer surveillance in the comments below. Please share this article with those who may find this research helpful.

