For decades, the human immune system has been locked in a sophisticated game of cat-and-mouse with Herpes simplex virus 1 (HSV-1). Whereas the virus is well-known for causing cold sores and occasional neurological complications, the molecular machinery the body uses to detect its presence—and the ways the virus tries to hide—is only now coming into sharp focus.
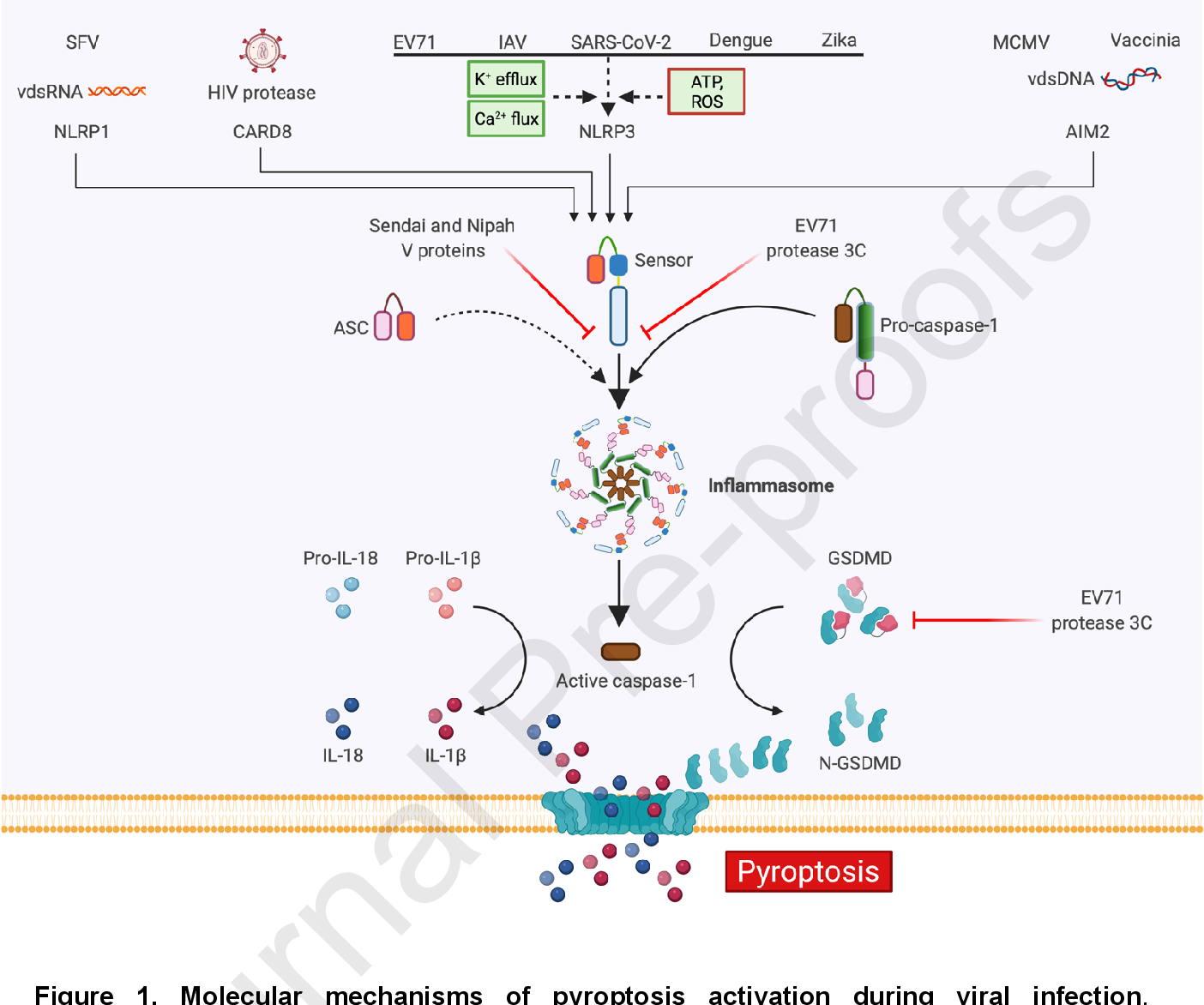
Recent research into innate immunity has identified a specific genomic “red flag” that allows the body to spot the intruder: poly(T) DNA sequences. These long stretches of thymine act as a primary trigger for the HSV-1 poly(T) DNA and AIM2 inflammasome activation, a critical defense mechanism that signals the cell to sound the alarm and, in many cases, undergo a programmed death to stop the virus from spreading.
As a physician, I identify this level of molecular precision fascinating. It transforms our understanding of the virus from a simple pathogen into a complex entity that carries its own “detection codes” within its genome. The discovery of poly(T) DNA as a key ligand for the AIM2 inflammasome provides a clearer map of how our innate immune sensors distinguish between our own genetic material and that of a foreign invader.
The Molecular Sentinel: How AIM2 Detects HSV-1
The AIM2 (Absent in Melanoma 2) protein serves as a molecular sentinel within the cell’s cytoplasm. Its primary job is to monitor for the presence of double-stranded DNA (dsDNA) that shouldn’t be there. Under normal conditions, human DNA is sequestered inside the nucleus or mitochondria; when AIM2 encounters dsDNA in the cytosol, it recognizes it as a sign of infection or cellular damage.
While AIM2 can bind to various forms of DNA, research indicates that poly(T) DNA sequences—repetitive stretches of the nucleotide thymine—are particularly potent ligands. When these sequences are detected, AIM2 undergoes a conformational change, recruiting other proteins to form a large complex known as the inflammasome. This complex activates caspase-1, an enzyme that triggers the release of pro-inflammatory cytokines and induces pyroptosis, a highly inflammatory form of programmed cell death.
This process is a cornerstone of host defense. By sacrificing the infected cell through pyroptosis, the body not only eliminates the viral factory but also releases signals that recruit other immune cells to the site of infection, effectively coordinating a localized counter-attack.
Viral Sabotage: The Role of VP22
HSV-1 does not succumb to this detection system without a fight. To ensure its survival and efficient replication, the virus has evolved a specialized tool for sabotage: the VP22 tegument protein. VP22 acts as a shield, attempting to neutralize the AIM2 response before the alarm can be fully raised.
Studies have shown that VP22 can inhibit the activation of the AIM2 inflammasome, likely by interfering with the DNA-binding activity of the sensor. By blocking the interaction between the poly(T) DNA ligands and the AIM2 protein, the virus can essentially “blind” the cell’s sentinel, allowing it to replicate undetected for a longer period. This evasion tactic is a primary reason why HSV-1 is so effective at establishing latent infections in the human body.
The battle between the AIM2 sensor and the VP22 protein represents a critical tipping point in the infection. If the immune system detects the poly(T) sequences faster than VP22 can suppress them, the infection is contained. If the virus wins this initial skirmish, it can spread more effectively through the host’s tissues.
Beyond Simple Death: The Rise of PANoptosis
While pyroptosis is a powerful tool, the body often employs a more integrated strategy called PANoptosis. This is not a single pathway but a coordinated convergence of three distinct types of programmed cell death: pyroptosis, apoptosis, and necroptosis.
Research suggests that AIM2 does not operate in isolation. Instead, it can form part of a larger molecular platform called the PANoptosome. In this complex, AIM2 collaborates with other sensors, such as ZBP1 (Z-DNA binding protein 1) and pyrin, to ensure that if one pathway of cell death is blocked by the virus, another can take its place.
The synergy of these pathways provides a failsafe mechanism. If HSV-1 manages to inhibit caspase-1 (the driver of pyroptosis), the PANoptosome can shift the cell toward apoptosis or necroptosis. This plasticity makes it significantly harder for the virus to evade the host’s defenses entirely.
Comparing Immune Responses to HSV-1
| Component | Role in Defense | Viral Counter-Measure |
|---|---|---|
| Poly(T) DNA | Acts as the ligand/trigger for detection | Sequestration or shielding |
| AIM2 Protein | Senses cytosolic DNA and forms inflammasome | Inhibition via VP22 protein |
| Caspase-1 | Triggers pyroptosis and cytokine release | Protease inhibition |
| PANoptosome | Coordinates multiple cell death pathways | Complex evasion strategies |
Why This Matters for Future Medicine
Understanding the specific interaction between poly(T) DNA and the AIM2 inflammasome opens new doors for therapeutic intervention. If we can develop tiny molecules that prevent VP22 from blocking AIM2, we could potentially “unmask” the virus, making it far more susceptible to the body’s natural defenses.
this research informs the development of next-generation vaccines. By understanding which genomic sequences—like the poly(T) stretches—are most effective at triggering a robust innate response, scientists can better design adjuvants or synthetic antigens that prime the immune system to recognize and destroy the virus more efficiently upon first exposure.
The broader implications extend to other DNA viruses as well. The principles of cytosolic DNA sensing and the utilize of repetitive sequences as ligands are likely shared across various viral families, meaning the lessons learned from HSV-1 could help us combat a wide array of infectious diseases.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
The next phase of research is expected to focus on the structural mapping of the AIM2-poly(T) complex using high-resolution cryo-electron microscopy. This will allow researchers to see exactly how the “key” fits into the “lock,” potentially leading to the design of highly specific immune-boosting drugs. We await further peer-reviewed data on how these mechanisms vary across different human genotypes, which may explain why some individuals are more prone to severe herpes outbreaks than others.
Do you have questions about the latest in immunology or viral research? Share your thoughts in the comments or share this article with your network to keep the conversation going.