For years, Jenny Keepe, a 34-year-old woman from Warrington, believed she was simply managing her daily appearance. She had a routine of wearing long artificial eyelashes, a beauty choice that inadvertently masked a life-threatening medical condition. It was only after she removed the extensions during the COVID-19 pandemic that a subtle but critical physical change became visible: her right eye had begun to protrude.
What initially seemed like a minor cosmetic anomaly was, in reality, the only outward sign of a non-malignant brain tumor known as a meningioma. The discovery sparked a medical emergency that led to urgent surgery and a subsequent investigation into the potential cause of the growth—a long-term reliance on a specific form of injectable contraception.
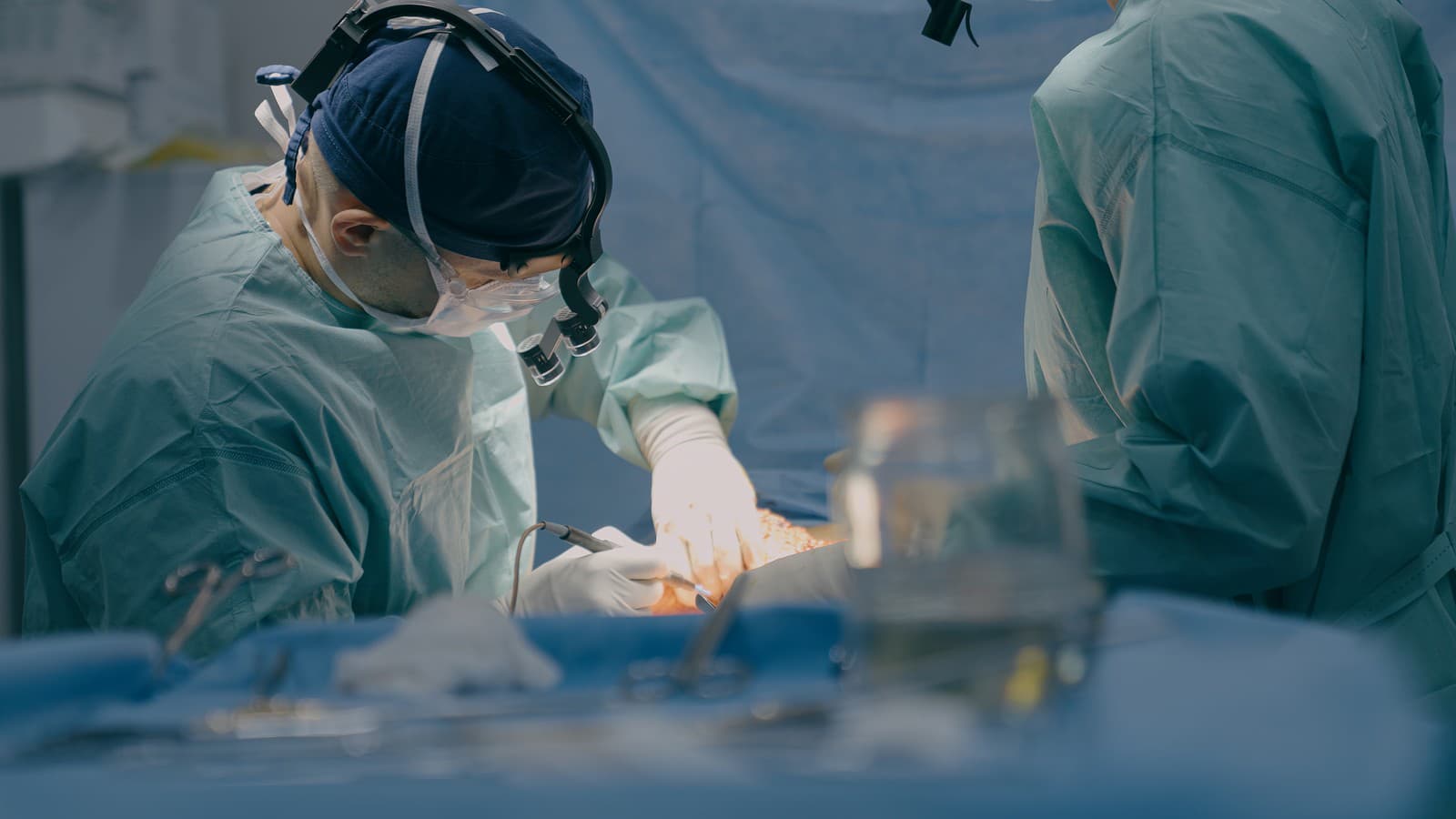
Keepe’s experience serves as a stark reminder of how easily subtle symptoms can be overlooked and the complexities of long-term medication side effects. After a family gathering in 2020, her mother noticed the asymmetry in her eyes. Despite Keepe’s initial dismissal of the observation, the discovery led to an optician’s visit where a suspicious area was detected behind the right eye. Within a week, an emergency MRI confirmed the presence of the tumor.
The Link Between Medroxyprogesterone and Meningiomas
Following her diagnosis, medical professionals pointed toward Keepe’s history with injectable contraception containing medroxyprogesterone acetate (MPA). Having used the medication since the age of 17, Keepe was informed that long-term exposure to high doses of this synthetic progestogen is associated with the development of meningiomas.

The shock of the diagnosis was compounded by the lack of traditional warning signs. Keepe noted that she had experienced no headaches or other typical neurological symptoms. To address the growth, she underwent two surgeries to remove approximately 50 percent of the tumor, followed by radiotherapy. Her condition is currently classified as stable, though she remains under annual medical surveillance to monitor for any further growth.
Keepe has expressed significant frustration over the lack of transparency regarding the risks associated with the injection.
“These things didn’t have to happen. I was never told about the risks,”
she stated, adding that had she been informed, she would have ceased using the medication years earlier to avoid the risk of a stroke or seizure.
Regulatory Responses and Patient Safety
The medical community and regulatory bodies have acknowledged the connection between high-dose MPA and brain tumors, though they emphasize that the absolute risk remains low. In October 2024, the pharmaceutical company Pfizer, in coordination with the Medicines and Healthcare products Regulatory Agency (MHRA), noted a slight increase in the risk of meningioma associated with long-term, high-dose use of medroxyprogesterone.
The National Institute for Health and Care Excellence (NICE) has similarly listed meningioma as a potential side effect of high-dose, long-term use and recommends the discontinuation of the drug if such a tumor is diagnosed. The MHRA continues to monitor the safety of synthetic progestogens to ensure patient guidelines remain current.
Dr. Alison Cave emphasized that patient safety is the primary priority, noting that drug information was updated in 2024 following studies on MPA and meningioma risks. By early 2025, these updates were further expanded to include new risk management measures and updated patient leaflets, designed to help users recognize symptoms and contact their healthcare providers promptly.
Industry Perspective and Risk Assessment
Pfizer has stated This proves fully engaged with the European Medicines Agency (EMA) in evaluating these risks. While the company acknowledges a significantly increased risk for those using high doses over several years, it maintains that the overall risk for the general population is very small.
A spokesperson for Pfizer highlighted that medroxyprogesterone acetate has been approved in over 60 countries for three decades and has served millions of patients with a well-documented safety profile. The company has updated its product leaflets to reflect the potential link between high-dose usage and meningiomas, urging patients to consult their doctors if they have concerns.
| Period | Key Event |
|---|---|
| 2020 | Symptom identified by family; emergency MRI confirms tumor. |
| Post-Op | Patient advised to immediately cease medroxyprogesterone use. |
| Oct 2024 | Pfizer and MHRA report increased risk for high-dose MPA users. |
| Early 2025 | Updated patient leaflets and risk management measures implemented. |
For those currently using injectable contraception, medical professionals recommend regular check-ups and a transparent dialogue with providers about the duration of use. Keepe advises other women to consider the possibility of switching methods after several years of use to mitigate potential long-term risks.
Disclaimer: This article is provided for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or medication.
The MHRA and EMA will continue their ongoing monitoring of synthetic progestogens, with further safety updates expected as more long-term data becomes available through 2025.
We invite you to share your thoughts or experiences with healthcare transparency in the comments below.

