Researchers at the National Cancer Center (NCC) in Korea have uncovered a critical molecular mechanism that allows acute lymphoblastic leukemia (ALL) cells to thrive by accelerating fatty acid synthesis. The discovery, led by Dr. Jung-hyun Kim of the Cancer Metastasis Research Division, provides a fresh understanding of how these aggressive blood cancer cells fuel their own rapid growth and survival.
Acute lymphoblastic leukemia is a fast-growing cancer of the white blood cells that occurs most commonly in children, though it likewise affects adults. By identifying the specific molecular pathway that triggers lipid production, the research team has highlighted a potential vulnerability in the cancer’s metabolic machinery that could be targeted to stall disease progression.
The study reveals that ALL cells do not merely rely on external nutrients but actively reprogram their internal chemistry to synthesize the fats they need for membrane construction and energy. This metabolic shift is essential for the proliferation of malignant cells, making the leukemia growth mechanism a primary target for future therapeutic intervention.
The Role of Fatty Acid Synthesis in Cancer Progression
In healthy cells, fatty acid synthesis is tightly regulated to maintain homeostasis. But, in acute lymphoblastic leukemia, this balance is disrupted. Dr. Kim’s team identified that the cancer cells hijack specific molecular signals to ramp up the production of lipids, which act as the building blocks for new cell membranes during rapid division.
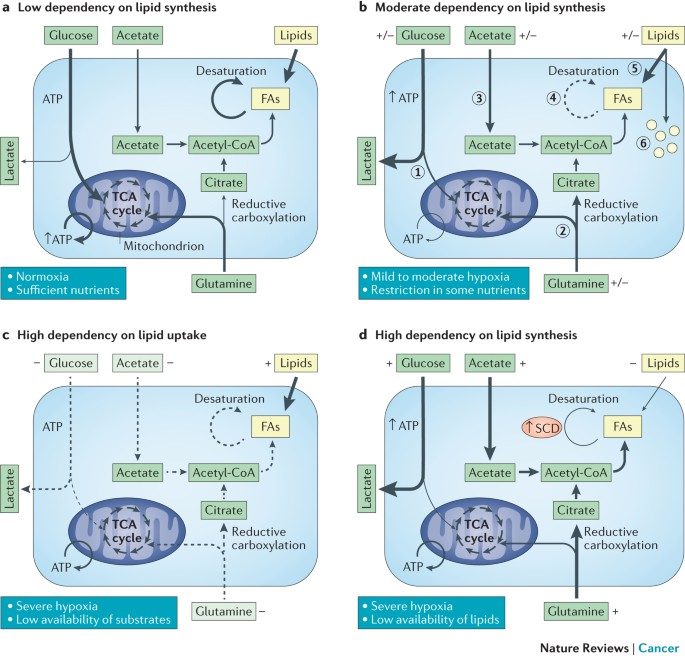
This process is not merely a byproduct of cancer growth but a requirement. Without the ability to synthesize these fatty acids, the leukemia cells struggle to maintain their structural integrity and lose their ability to divide efficiently. The research underscores a fundamental shift in how we view the “diet” of a cancer cell, moving from a general need for glucose to a specific requirement for synthesized lipids.
The findings align with a broader trend in oncology research focusing on metabolic reprogramming. Many aggressive tumors exhibit a “metabolic addiction,” where they become dependent on a specific nutrient or pathway. In the case of ALL, the reliance on the fatty acid synthesis pathway creates a metabolic bottleneck that researchers can now potentially exploit.
From Molecular Discovery to Clinical Potential
The significance of this research lies in its potential to move toward “metabolic therapy.” Rather than relying solely on cytotoxic chemotherapy, which attacks all rapidly dividing cells, targeting the specific enzymes responsible for fatty acid synthesis could offer a more precise way to starve the cancer cells while sparing healthy tissue.
The research team focused on the molecular triggers that activate these lipid-producing pathways. By understanding the “on-switch” for fatty acid synthesis, scientists can now investigate pharmacological inhibitors—drugs designed to flip that switch off. This approach aims to reduce the tumor burden and potentially increase the efficacy of existing treatments by making the cells more fragile.
Key Implications for Patient Care
- Targeted Therapy: The discovery opens the door for the development of inhibitors that specifically block lipid synthesis in leukemia cells.
- Biomarker Identification: Measuring the levels of fatty acid synthesis markers could support clinicians predict how aggressive a particular case of ALL might be.
- Combination Strategies: Combining metabolic inhibitors with standard chemotherapy may prevent the cancer from developing resistance.
Understanding the Impact on ALL Patients
For patients diagnosed with acute lymphoblastic leukemia, the stakes are high due to the speed at which the disease progresses. The National Cancer Center‘s focus on the molecular drivers of growth is aimed at reducing the relapse rates that often plague survivors of the disease.
While the research is currently in the molecular and cellular phase, it provides the necessary blueprint for preclinical trials. The transition from a laboratory discovery to a bedside treatment typically involves validating the target in animal models and then proceeding to human clinical trials to ensure safety and efficacy.
| Feature | Healthy Leukocytes | ALL Cells (Research Findings) |
|---|---|---|
| Lipid Production | Regulated/Baseline | Highly Accelerated |
| Growth Rate | Controlled | Rapidly Proliferative |
| Energy Source | Balanced Nutrients | Dependent on Fatty Acid Synthesis |
| Therapeutic Target | General Homeostasis | Specific Lipid Synthesis Pathways |
The Broader Landscape of Hematologic Oncology
This breakthrough is part of a larger global effort to decode the “metabolome” of cancer. Institutions worldwide are increasingly recognizing that the way a cell processes energy is just as important as the genetic mutations it carries. By mapping the molecular principles of how ALL cells grow, the NCC team is contributing to a global database of cancer vulnerabilities.
The complexity of leukemia means that a “one size fits all” approach rarely works. The identification of this specific growth secret allows for a more personalized approach to medicine, where a patient’s specific metabolic profile could dictate the choice of therapy. This shift toward precision medicine is the current gold standard in oncology, aiming to maximize patient survival while minimizing the debilitating side effects of systemic chemotherapy.
For more detailed information on cancer research and current clinical trials, patients and caregivers are encouraged to visit the National Cancer Institute or official government health portals.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Patients should consult with their primary physician or a board-certified oncologist for diagnosis and treatment options.
The next phase for this research involves the identification of specific little-molecule inhibitors that can effectively block the identified fatty acid synthesis pathway in vivo. Updates on these developments are expected as the research team moves toward preclinical validation and potential partnership with pharmaceutical developers.
We invite readers to share their thoughts on the future of metabolic cancer therapy in the comments below and share this update with those who may find it helpful.

