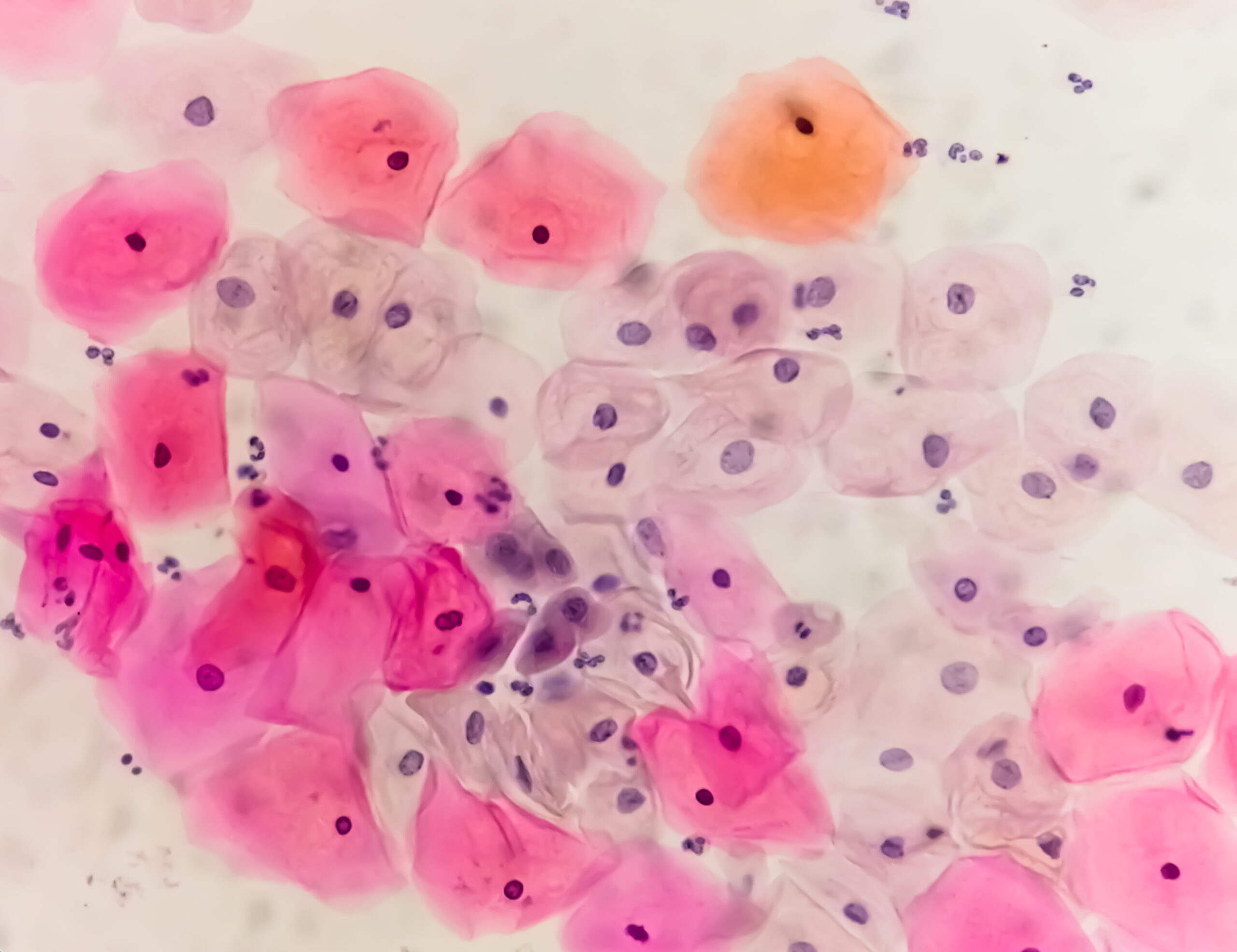
For years, the medical community has known that high-risk Human Papillomavirus (HR-HPV) is the primary driver of cervical cancer. Though, the great clinical challenge has never been simply detecting the virus—most women will clear an HPV infection on their own—but predicting which infections will progress into cervical intraepithelial neoplasia (CIN), the precancerous lesions that require medical intervention.
Novel research suggests that the secret to this prediction may lie in the vaginal microbiome, specifically the balance of beneficial bacteria known as lactobacilli. A recent study indicates that a decline in these bacteria, combined with specific biological markers, can help clinicians identify women at a significantly higher risk of developing CIN, potentially transforming how we manage HPV-positive patients.
As a physician and medical writer, I have followed the evolving science of the microbiome for years. We are moving away from a “one size fits all” approach to screening and toward a more personalized model of preventive care. By analyzing the interplay between vaginal lactobacilli and high-risk HPV, researchers are developing tools that move beyond the binary “positive or negative” HPV test to a more nuanced risk profile.
The Microbiome Connection: Why Lactobacilli Matter
The vaginal environment is a delicate ecosystem. In a healthy state, lactobacilli dominate the flora, producing lactic acid that maintains a low, acidic pH. This acidity acts as a natural defense mechanism, inhibiting the growth of pathogenic bacteria and potentially interfering with the ability of HPV to integrate into the cervical epithelium.
When this balance shifts—a state often characterized by a decrease in lactobacilli and a subsequent rise in vaginal pH—the cervical lining becomes more vulnerable. The study analyzed 358 women infected with HR-HPV to determine which factors most strongly correlated with the presence of CIN. The findings revealed that a diminished population of lactobacilli was not just a byproduct of infection, but an independent risk factor for the development of precancerous lesions.
This biological vulnerability is further compounded by other factors. The research identified a clear correlation between CIN and an increased HR-HPV viral load, as well as the duration of the persistent infection. The longer the virus remains active in an environment with low lactobacilli levels, the higher the likelihood of cellular transformation.
Identifying the High-Risk Profile
The researchers didn’t stop at bacteria. They looked at a broader array of patient data to create a comprehensive risk map. Interestingly, the study pointed to a surprising systemic factor: serum Vitamin D levels. A lower concentration of Vitamin D in the blood was identified as an independent risk factor for CIN, likely due to the vitamin’s role in modulating the immune response and maintaining mucosal integrity.

To make this data actionable for doctors, the team developed a “nomogram”—a sophisticated clinical prediction tool. By plugging in several variables, a clinician can calculate a patient’s specific probability of having CIN. The model was tested across two groups: a training group of 251 women and a validation group of 107 women, showing consistent accuracy in both.
| Risk Factor | Correlation with CIN | Biological Impact |
|---|---|---|
| Vaginal Lactobacilli | Decreased | Loss of acidic barrier and immune support |
| Vaginal pH | Increased | Creates a more hospitable environment for HPV |
| HPV Viral Load | Increased | Higher intensity of viral activity in cells |
| Serum Vitamin D | Decreased | Weakened mucosal immunity |
| Infection Duration | Increased | Higher chance of genomic integration |
Measuring the Accuracy of the Prediction Tool
In medical research, we look at the “AUC” (Area Under the Curve) and the “C-index” to determine if a tool is actually useful in a clinic. An AUC of 0.5 is no better than a coin flip, whereas 1.0 is perfect. This nomogram achieved an AUC of 0.845 in the training set and 0.761 in the validation set, indicating a strong predictive capability.

The sensitivity and specificity of the model—essentially its ability to correctly identify those with the disease and correctly rule out those without it—were reported at 0.660 and 0.845 in the training group and 0.619 and 0.725 in the validation group. While not perfect, these figures suggest that the tool can significantly narrow down which patients need immediate, invasive biopsies and which can be safely monitored.
For the patient, this means fewer unnecessary procedures. For the healthcare system, it means a more efficient allocation of resources, focusing intensive interventions on the women whose biological profiles—marked by low lactobacilli and low Vitamin D—put them at the highest risk.
The Path Toward Targeted Intervention
The implications of this research extend beyond mere prediction. If we recognize that a lack of lactobacilli and low Vitamin D correlate with CIN progression, it opens the door for targeted preventive strategies. While the study focuses on prediction, future clinical applications may explore whether restoring the vaginal microbiome or correcting Vitamin D deficiencies could slow the progression of HPV-related lesions.
Current guidelines from organizations like the World Health Organization emphasize a comprehensive approach to cervical cancer elimination, including vaccination and screening. Integrating microbiome analysis into this framework could provide the “missing link” in understanding why some women progress to cancer while others do not.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Patients should consult with a licensed healthcare provider for diagnosis and treatment of HPV or CIN.
The next step for this research will likely involve larger, prospective longitudinal studies to determine if the nomogram’s predictions hold true across more diverse populations over several years. As these tools move from the research phase to clinical trials, they may soon become a standard part of the gynecological exam for women with persistent high-risk HPV.
Do you reckon microbiome testing should become a standard part of cervical cancer screening? Share your thoughts in the comments or share this article with your network.