For years, patients with seronegative myasthenia gravis (MG) have lived in a diagnostic shadow. While those with the more common forms of the disease could point to a positive blood test for specific antibodies as a “smoking gun,” seronegative patients—who lack these detectable markers—often faced a grueling “diagnostic odyssey.” They experienced the hallmark symptoms of MG—drooping eyelids, slurred speech, and profound muscle weakness—yet their lab results remained stubbornly normal, leaving them to wonder if their symptoms were real or if their doctors were simply missing something.
That ambiguity is shifting with the latest clinical validation and FDA-supported application of efgartigimod (brand name Vyvgart). By demonstrating significant efficacy in the ADAPT SERON trial, efgartigimod is establishing itself as a critical tool for those who have historically been the hardest to diagnose and treat. For the first time, there is a targeted therapy with robust evidence specifically for the seronegative population, moving these patients from a state of clinical guesswork to a standardized path of care.
As a physician, I have seen how the label “seronegative” can inadvertently lead to delays in treatment. In myasthenia gravis, the immune system mistakenly attacks the communication points between nerves and muscles. In most patients, we can see the antibodies responsible for this attack. In seronegative patients, the antibodies are either absent, too few to detect, or of a variety that current tests cannot identify. This does not mean the disease isn’t there. it simply means the traditional “map” used by clinicians doesn’t work.
The Challenge of the “Invisible” Diagnosis
Myasthenia gravis is often called “the snowflake disease” because it manifests differently in every patient. However, the distinction between seropositive and seronegative MG is the most significant dividing line in clinical management. Most patients test positive for acetylcholine receptor (AChR) antibodies or muscle-specific kinase (MuSK) antibodies. When these are absent, the patient is classified as seronegative.

The stakes for this classification are high. Without a positive antibody test, clinicians often rely on more invasive tests, such as single-fiber electromyography (SFEMG) or repetitive nerve stimulation, which are not available at every clinic. This gap in diagnostics often leads to a delay in starting immunosuppressive therapy, allowing muscle weakness to progress and decreasing the patient’s overall quality of life.
The approval and application of efgartigimod for this group represents a paradigm shift. It suggests that the underlying mechanism of the disease—the accumulation of harmful IgG antibodies—is present and treatable even when those antibodies evade standard laboratory detection.
How Efgartigimod Rewrites the Immune Response
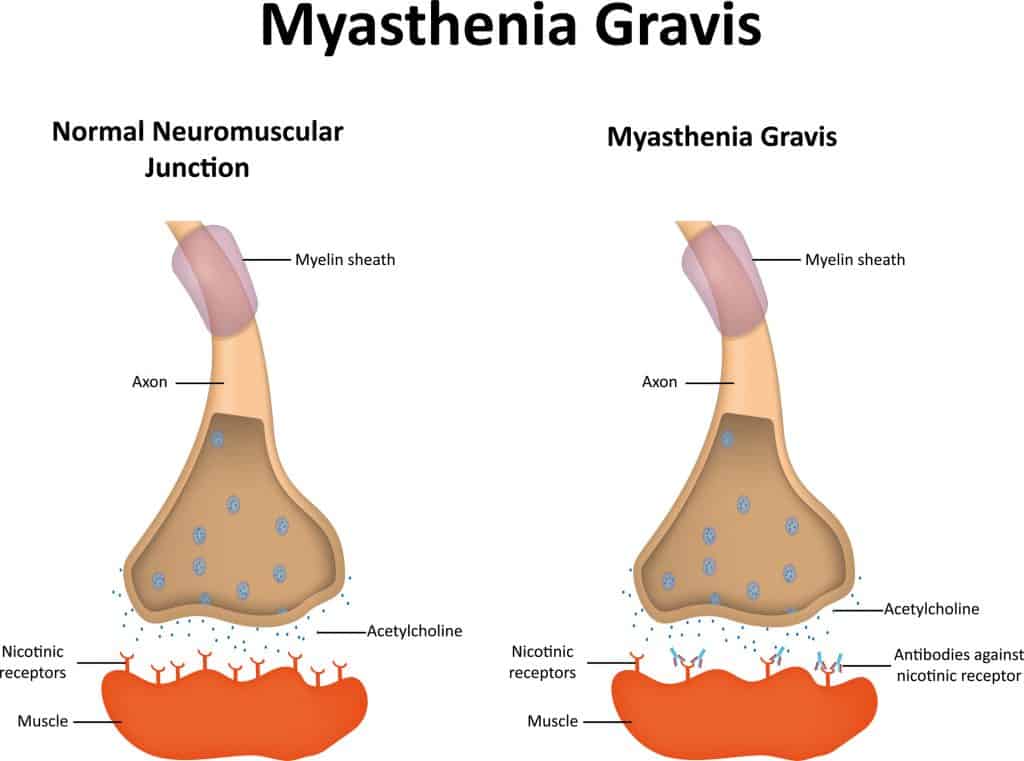
To understand why efgartigimod works for seronegative patients, one must look at the neonatal Fc receptor (FcRn). In a healthy body, FcRn acts as a recycling center for IgG antibodies; it binds to them and prevents them from being broken down and cleared from the bloodstream.
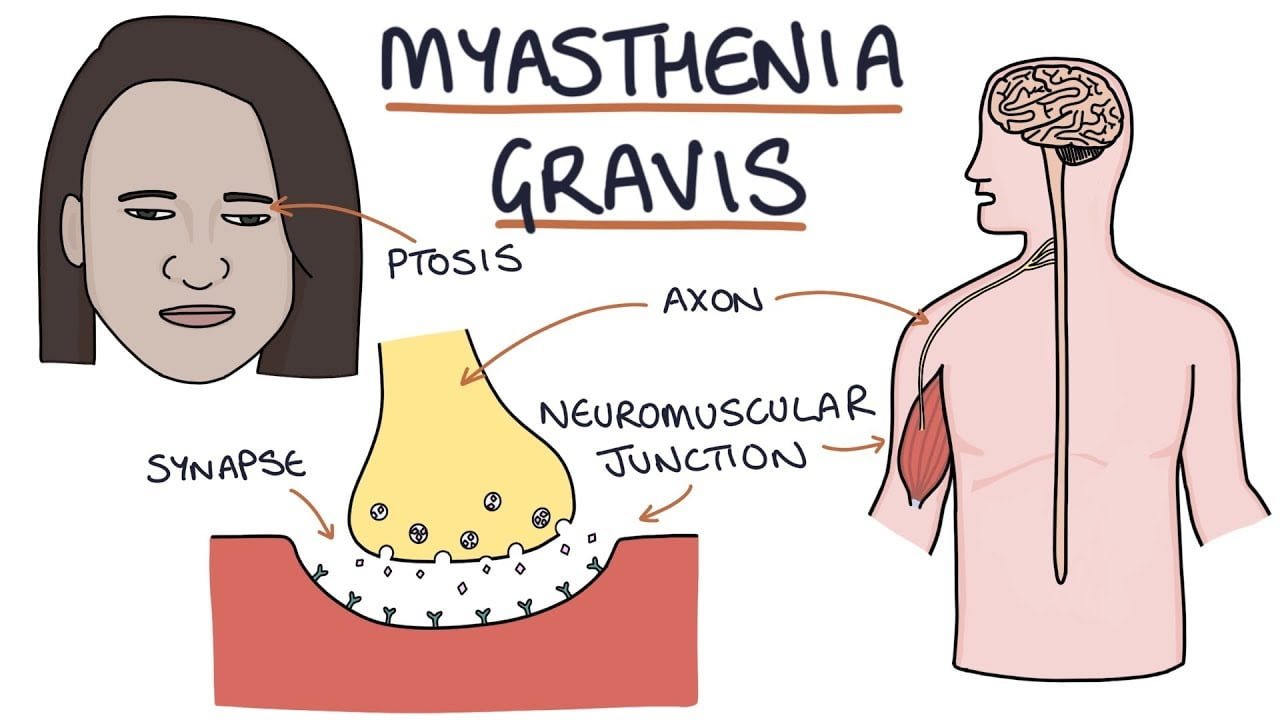
In patients with MG, the body produces pathogenic IgG antibodies that block or destroy the receptors at the neuromuscular junction. Efgartigimod is a human IgG1 antibody fragment that blocks the FcRn receptor. By “clogging” this recycling center, the drug forces the body to stop recycling these harmful antibodies, allowing them to be cleared naturally and more rapidly from the system.
The brilliance of this mechanism is that it is “antibody-agnostic.” It doesn’t matter if the antibody is AChR, MuSK, or a rare, unidentified variant found in seronegative patients; as long as the pathology is driven by IgG antibodies, efgartigimod can reduce their concentration. This makes it a precision tool that functions regardless of the patient’s serostatus.
Breaking Down the ADAPT SERON Evidence
The clinical foundation for this advancement is the ADAPT SERON trial (NCT06298552), which specifically enrolled 119 patients with seronegative MG. The study was designed to determine if the benefits seen in the broader generalized MG (gMG) population translated to those without detectable antibodies.
The primary endpoint of the study was the change in the Myasthenia Gravis Activities of Daily Living (MG-ADL) score. What we have is a validated patient-reported outcome that measures the ability to perform basic tasks such as chewing, talking, brushing teeth, and breathing. The results were definitive: patients receiving four weekly IV infusions of efgartigimod showed a significant improvement in their MG-ADL scores compared to those receiving a placebo.
| Feature | Seropositive MG | Seronegative MG |
|---|---|---|
| Antibody Markers | AChR or MuSK positive | No detectable AChR/MuSK |
| Diagnosis Speed | Typically faster via blood test | Often delayed; requires SFEMG |
| Mechanism | IgG-mediated attack | IgG-mediated (often unidentified) |
| Efgartigimod Action | Reduces IgG via FcRn block | Reduces IgG via FcRn block |
A New Standard for Patient Care
The impact of this development extends beyond the pharmacy. For neurologists, the ADAPT SERON data provides a validated clinical pathway for patients who previously fell into a “gray area” of treatment. Instead of relying solely on broad-spectrum corticosteroids—which carry heavy side effects like weight gain, insomnia, and bone loss—physicians now have a targeted option that addresses the root cause of the weakness.
For the patients, the benefit is measured in autonomy. An improvement in MG-ADL scores isn’t just a data point on a chart; it is the difference between needing assistance to swallow food and eating independently, or the ability to hold a conversation without losing breath.
However, constraints remain. Access to these therapies often depends on insurance coverage and the availability of infusion centers. While efgartigimod is highly effective, it is not a cure; it is a management strategy that requires ongoing administration to maintain the reduction of antibody levels.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Patients should consult with a licensed healthcare provider for diagnosis and treatment options.
The next confirmed checkpoint for this therapy involves the continued monitoring of long-term safety and efficacy data from the ADAPT SERON extension phases, which will determine the optimal maintenance dosing for seronegative patients over several years.
Do you or a loved one navigate the challenges of a seronegative diagnosis? Share your experience in the comments or share this article to help others find the information they need.

