For decades, digoxin has been one of the most polarizing medications in the cardiology toolkit. Derived originally from the foxglove plant, the drug is prized for its ability to strengthen heart contractions and control heart rate, yet it is often viewed with caution due to a narrow therapeutic window—the slim margin between a helpful dose and a toxic one.
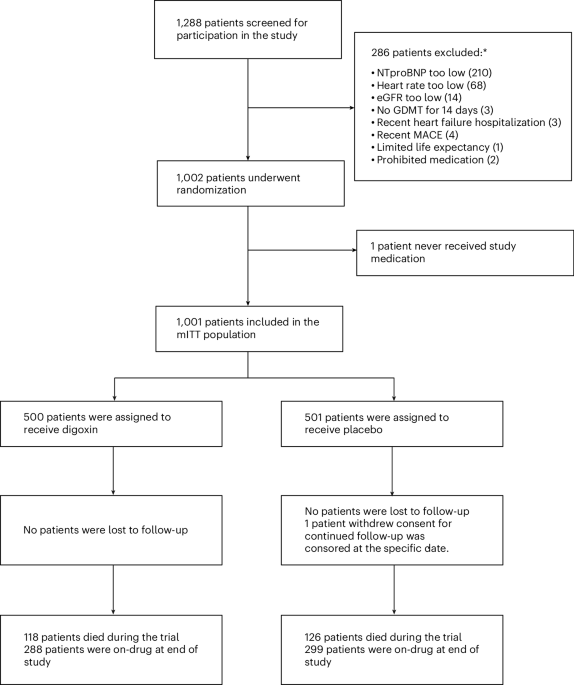
As modern medicine introduces a wave of sophisticated, expensive new therapies for heart failure, questions have mounted over whether this old mainstay still has a place in the clinic. The DECISION trial, a randomized controlled study focusing on patients with reduced or mildly reduced ejection fraction (HFrEF and HFmrEF), sought to determine if adding low-dose digoxin to current gold-standard treatments could further reduce the risk of cardiovascular death and worsening heart failure.
The results, while not reaching the threshold of statistical significance for its primary goal, offer a nuanced perspective on the drug’s utility. While low-dose digoxin did not significantly lower the composite rate of cardiovascular death and heart failure events compared to a placebo, the study confirmed that when administered carefully, the drug is remarkably safe and well-tolerated, even in populations previously thought to be at higher risk.
For clinicians and patients, the takeaway is not that digoxin is obsolete, but that its value may lie in its accessibility and safety profile when integrated into a modern treatment regimen.
Understanding the “Missing” Statistical Significance
In the world of clinical trials, a “non-significant” result is often misinterpreted as a “failed” result. However, the DECISION trial reveals a more complex story. The researchers observed that the absolute number of primary outcome events was actually lower in the digoxin group than in the placebo group. The relative risk reduction was 19%, just shy of the 22% reduction the researchers had projected when calculating their sample size.
Several real-world factors likely muted the drug’s statistical signal. Most notably, the trial coincided with the peak of the COVID-19 pandemic, beginning in July 2020. During this window, hospital visits were restricted, and many follow-ups were shifted to telephone calls, leading to a higher-than-expected rate of patients discontinuing the study medication.
Age also played a role. With a mean patient age of 72, the DECISION cohort was significantly older than those in previous landmark trials like DIG or DIGIT-HF. Elderly patients are more likely to stop medications due to the complexities of polypharmacy—the use of multiple drugs—or the natural progression of their disease.
the landscape of heart failure treatment shifted during the trial. A higher proportion of patients in the placebo arm received sodium-glucose cotransporter-2 (SGLT2) inhibitors, a newer class of drugs that has revolutionized heart failure care. This “up-titration” of other guideline-recommended therapies may have closed the gap between the placebo and digoxin groups, making the specific benefit of digoxin harder to isolate.
Correcting the Record for Women
One of the most significant contributions of the DECISION trial is its data on women. Historically, some post hoc analyses—specifically from the 2002 DIG study—suggested that women might face an increased risk of death when taking digoxin. However, those earlier findings were linked to higher serum digoxin concentrations (SDC) in women compared to men.
The DECISION trial utilized a precise dosing algorithm to keep patients within a target range of 0.5 to 0.9 ng/ml. Under these controlled conditions, low-dose digoxin proved safe for the 284 women in the study. This aligns with previous sub-analyses suggesting that the “danger” associated with digoxin in women was not a result of the drug itself, but rather a result of dosing that exceeded the therapeutic window.
By demonstrating that low-dose digoxin is safe for women when monitored correctly, the study provides critical reassurance to physicians who may have previously avoided the drug in female patients based on outdated or misinterpreted data.
A Safety Net for Atrial Fibrillation
The DECISION trial also fills a critical gap in the literature regarding patients with atrial fibrillation (AF), a common comorbidity in heart failure that complicates heart rate management. While the earlier DIG trial focused exclusively on patients in sinus rhythm, DECISION is now the largest randomized, placebo-controlled outcome study to investigate low-dose digoxin in patients with AF.
The study found that adding digoxin to beta-blockers was safe and showed efficacy similar to that seen in patients without AF. This is a vital finding because the safety of digoxin in AF patients has often been questioned based on retrospective, non-randomized data, which is less reliable than the gold-standard randomized controlled trial (RCT) format.
When viewed alongside the DIGIT-HF trial—which used digitoxin and showed a 15% lower combined risk of hospitalization or death—the evidence suggests that digitalis glycosides remain a viable option for stabilizing patients with complex heart rhythms and reduced heart function.
Comparing the Digitalis Landmark Trials
| Trial | Primary Focus | Key Finding | Patient Population |
|---|---|---|---|
| DIG | Worsening HF events | 28% relative risk reduction | Sinus rhythm |
| DIGIT-HF | Hospitalization/Death | 15% lower combined risk | Advanced heart failure |
| DECISION | CV Death/Worsening HF | Non-significant (19% reduction) | HFrEF/HFmrEF & AF |
The Practical Path Forward
While the “blockbuster” statistical result may have been elusive, the clinical utility of low-dose digoxin remains. For a significant portion of the population—specifically those with a mildly reduced ejection fraction (LVEF >40%)—the drug provided a consistent safety profile without increasing the need for pacemaker implantations or causing severe adverse effects.

In an era of prohibitively expensive specialty drugs, digoxin remains a simple, cheap, and effective tool. For patients who continue to experience symptoms despite guideline-directed medical therapy, or for those whose atrial fibrillation requires tighter rate control, low-dose digoxin offers a low-cost intervention with a now-well-documented safety profile.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Patients should always consult with a healthcare provider before starting or changing any medication regimen.
The medical community now looks toward the integration of these findings into updated clinical guidelines for heart failure. Future updates from the European Society of Cardiology (ESC) and the American Heart Association (AHA) will likely reflect this totality of evidence, potentially refining the specific patient profiles—such as those with AF or HFmrEF—who benefit most from low-dose digitalis therapy.
Do you or a loved one manage heart failure? We want to hear about your experience with these treatments. Share your thoughts in the comments below or share this article with others who may find it helpful.