Understanding how the body responds to new treatments is a complex process, particularly in rare diseases. Recent discussion surrounding the use of iptacopan, a medication showing promise for C3 glomerulopathy (C3G), has highlighted the importance of carefully interpreting changes in complement biomarkers – substances that indicate activity of the complement system, a part of the immune system. Researchers involved in the pivotal APPEAR-C3G trial are clarifying key points about these biomarkers and their relationship to treatment response, aiming to refine how these indicators are used in clinical practice.
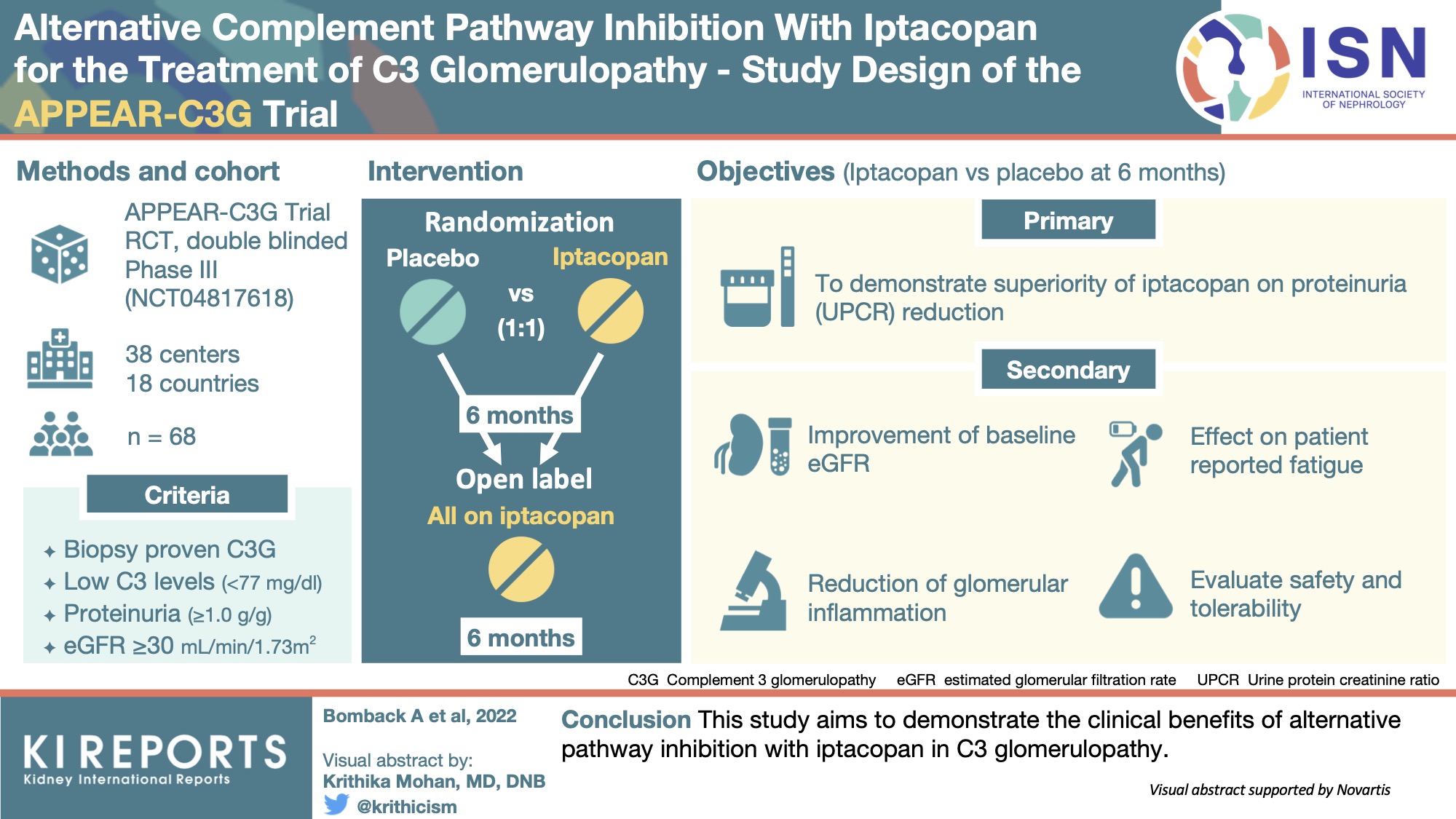
C3 glomerulopathy is a rare and serious kidney disease caused by dysregulation of the complement system. The condition can lead to kidney failure, requiring dialysis or transplantation. Iptacopan works by blocking a key enzyme in the complement cascade, aiming to reduce the damaging inflammation in the kidneys. The APPEAR-C3G trial, published in the New England Journal of Medicine in 2023, demonstrated significant reductions in proteinuria – protein in the urine, a key marker of kidney damage – in patients treated with iptacopan.
The recent exchange stems from commentary on the original study, specifically focusing on how changes in complement biomarkers should be interpreted during iptacopan treatment. The authors of the APPEAR-C3G trial emphasize that the expected biomarker changes don’t always align with clinical improvement, and a simple “on-treatment” versus “off-treatment” interpretation can be misleading. The goal is to move toward a more nuanced understanding of how these biomarkers reflect the drug’s impact on the disease process.
The Nuances of Complement Biomarker Interpretation
One key point of clarification concerns factor B, a protein involved in the alternative pathway of the complement system. Researchers observed that factor B levels often increase with iptacopan treatment, even as the drug effectively blocks the pathway and reduces proteinuria. This seemingly paradoxical finding is due to iptacopan’s mechanism of action. By blocking the pathway, the drug prevents the consumption of factor B, leading to its accumulation in the blood. An increase in factor B doesn’t necessarily indicate treatment failure; it can actually be a sign that the drug is working as intended.
Similarly, changes in other complement biomarkers, such as C3a and C5a – fragments released during complement activation – require careful consideration. While iptacopan is designed to reduce the production of these fragments, their levels can fluctuate due to various factors, including individual patient variability and the presence of other inflammatory processes. The trial data showed that reductions in these biomarkers didn’t always correlate directly with improvements in proteinuria, highlighting the need for a more holistic assessment of treatment response.
The researchers stress that clinical outcomes, such as changes in proteinuria and estimated glomerular filtration rate (eGFR – a measure of kidney function), should remain the primary indicators of treatment success. Biomarkers are valuable tools for understanding the underlying disease mechanisms and monitoring treatment effects, but they should not be used in isolation to develop clinical decisions. The focus should be on whether the patient is experiencing a tangible benefit in terms of kidney function and reduced protein loss.
Context from the APPEAR-C3G Trial
The APPEAR-C3G trial involved 87 participants with C3G who were randomized to receive either iptacopan or placebo. The trial demonstrated that iptacopan led to a statistically significant and clinically meaningful reduction in proteinuria compared to placebo. Specifically, patients treated with iptacopan experienced a 44.2% reduction in urine protein-to-creatinine ratio (UPCR) from baseline at 28 weeks, compared to a 12.3% reduction in the placebo group. This difference was highly significant (p < 0.001), indicating a strong treatment effect.
the trial showed that iptacopan was generally well-tolerated, with a safety profile comparable to placebo. The most common adverse events were upper respiratory tract infections and headache, which were similar in both treatment groups. These findings support the potential of iptacopan as a safe and effective treatment option for patients with C3G.
The researchers acknowledge that further research is needed to fully elucidate the relationship between complement biomarkers and clinical outcomes in C3G. Ongoing studies are investigating the use of more sophisticated biomarker assays and exploring the potential of personalized treatment strategies based on individual biomarker profiles. The ultimate goal is to optimize treatment for each patient, maximizing the benefits of iptacopan while minimizing the risk of adverse effects.
What This Means for Patients with C3G
For individuals living with C3 glomerulopathy, this clarification is crucial. It emphasizes that treatment response isn’t always a straightforward equation. While biomarker monitoring remains key, patients and their doctors should prioritize clinical improvements – reductions in proteinuria and stabilization of kidney function – as the primary measures of success. Open communication with healthcare providers is essential to understand the nuances of biomarker interpretation and to tailor treatment plans accordingly.
The development of iptacopan represents a significant step forward in the treatment of C3G, a disease that has historically lacked effective therapies. The drug offers a new hope for patients who have previously faced limited options. However, it’s important to remember that iptacopan is not a cure, and ongoing monitoring and management are necessary to maintain long-term kidney health. The National Kidney Foundation provides comprehensive information about C3 glomerulopathy and available treatment options.
The researchers are continuing to analyze data from the APPEAR-C3G trial and other studies to refine their understanding of iptacopan’s effects on the complement system and its impact on C3G. Future research will focus on identifying biomarkers that can predict treatment response and on developing strategies to personalize treatment based on individual patient characteristics. The next key milestone will be the long-term follow-up data from the APPEAR-C3G trial, which will provide further insights into the durability of iptacopan’s effects and its impact on long-term kidney outcomes.
This evolving understanding of complement biomarkers and iptacopan treatment underscores the importance of ongoing research and collaboration in the field of nephrology. By working together, researchers and clinicians can continue to improve the lives of patients with C3 glomerulopathy and other rare kidney diseases.
Disclaimer: This article is for informational purposes only and should not be considered medical advice. Please consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
Have you or a loved one been affected by C3 glomerulopathy? Share your experiences and thoughts in the comments below. Please likewise share this article with anyone who might locate it helpful.