New restrictions on bone health monitoring in Ontario are sparking alarm among medical professionals and patient advocates, who warn that a “one-size-fits-all” approach to diagnostic testing could leave high-risk patients vulnerable to preventable fractures. As of April 1, 2026, the Ontario Health Insurance Plan (OHIP) implemented significant changes to the reimbursement of Bone Mineral Density (BMD) testing, fundamentally altering how often patients can receive these critical scans.
Under the new OHIP Schedule of Benefits, the timing for follow-up BMD tests is now dictated by provincial definitions of risk rather than the independent clinical judgment of a physician. Whereas baseline testing remains based on a provider’s assessment, subsequent monitoring intervals have been extended for the vast majority of patients, moving away from a system that previously allowed repeat testing every 12 months for high-risk individuals.
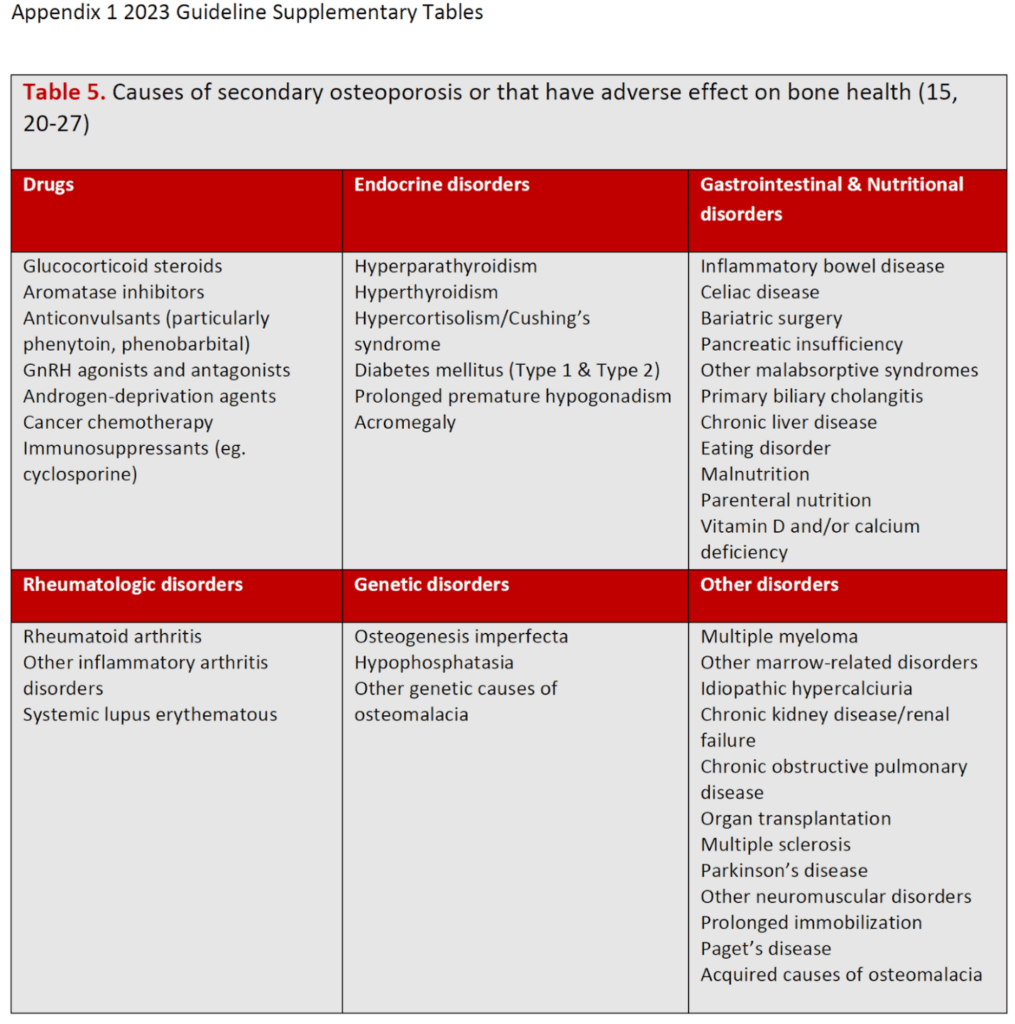
Osteoporosis Canada has raised urgent concerns that these changes in BMD testing in Ontario are inconsistent with the 2023 Clinical Practice Guideline for the Management of Osteoporosis in Canada. The organization argues that by ignoring critical caveats regarding “secondary causes” of bone loss, the province is restricting access to necessary care and potentially delaying the identification of rapid bone deterioration.
The shift has created immediate logistical chaos for clinics and hospitals, many of whom report receiving very little notice before the April 1 deadline. This has left healthcare providers scrambling to manage existing appointments, as tests already booked within the next 36 months may no longer be covered by the province, forcing patients to either postpone essential care or pay for the services out of pocket.
The New Risk Framework and the ’36-Month Gap’
The core of the controversy lies in how OHIP now defines “high risk.” The province is utilizing the Fracture Risk Assessment Tool (FRAX), or similar 10-year prediction tools, to determine eligibility for follow-up scans. According to the new guidelines, a patient is considered high risk if they have an estimated 10-year fracture risk of greater than 15%.
For these high-risk patients, OHIP now permits BMD monitoring only once every 36 months. This represents a stark departure from previous practice, where physicians could use their expertise to determine if a patient required a repeat test after 12 months. By shifting the authority from the bedside to a rigid schedule, critics argue that doctors have lost the ability to monitor treatment responses or disease progression effectively.
There is one narrow exception to the three-year rule. Repeat BMD testing every 12 months is still permitted for individuals with Hypercortisolism (Cushing’s syndrome) or those receiving high-dose glucocorticoid therapy, specifically Prednisone (or equivalent) at doses greater than 20 mg per day. But, medical advocates note that this specific prednisone threshold is higher than what current literature suggests as the trigger for significant bone loss.
| Patient Category | OHIP Permitted Interval | Clinical Concern |
|---|---|---|
| General High Risk (FRAX >15%) | Every 36 Months | Too long for rapid bone loss monitoring |
| Cushing’s / High-dose Prednisone | Every 12 Months | Prednisone threshold may be too high |
| Other Secondary Causes | Every 36 Months | Ignores guidelines for earlier reassessment |
| New Fragility Fractures | Every 36 Months | May delay urgent post-fracture monitoring |
Overlooked Risks and ‘Secondary Causes’
The tension between the provincial policy and clinical guidelines centers on a specific omission. While the 2023 Osteoporosis Canada Guideline does suggest a three-year interval for some, it includes a vital caveat: BMD testing should be reassessed earlier if a patient develops secondary causes, suffers a new fracture, or exhibits other risk factors for rapid bone loss.

By omitting this caveat, the OHIP schedule effectively ignores patients with hyperparathyroidism, rheumatoid arthritis, or genetic metabolic bone diseases. It likewise excludes those starting non-glucocorticoid medications associated with accelerated bone loss, such as androgen deprivation therapy or aromatase inhibitors. Patients transitioning off denosumab or anabolic bone-formation therapies—who are at an increased risk of rapid bone loss—are now forced into the 36-month window.
The impact is not limited to adults. Given that the 2023 guidelines focus on postmenopausal women and men over 50, children with secondary osteoporosis and fractures fall into a grey area where the rapid rate of bone growth may necessitate testing far more frequently than every three years.
Gender Disparity and Patient Implications
Because the risk of osteoporosis and fragility fractures is statistically higher in women, health advocates emphasize that these changes disproportionately impact women’s health. The restriction on testing creates a scenario where patients who genuinely require close monitoring are left vulnerable to “preventable and potentially significant bone loss.”
The practical implications for the Ontario healthcare system are three-fold:
- Patient Vulnerability: Delays in testing mean delays in treatment, increasing the likelihood of debilitating fractures.
- Clinical Constraint: Physicians are forced into a “one-size-fits-all” approach, stripping them of the clinical judgment needed to tailor care to the individual.
- Financial Burden: Patients who cannot wait three years for a scan may be forced to pay out of pocket for private testing.
Disclaimer: This article is provided for informational purposes only and does not constitute medical advice. Please consult a licensed healthcare provider for diagnosis and treatment planning.
The Path Toward Policy Revision
In response to the implementation of these rules, Osteoporosis Canada is currently coordinating a multi-pronged advocacy strategy. The organization’s Scientific Advisory Council has established contact with the Ontario Medical Association and the Ontario Association of Radiology to align their efforts in challenging the current Schedule of Benefits.
The primary objective of these groups is to secure a deferral of the changes until evidence-based expert advice is fully integrated into the OHIP policies. They are specifically calling for the 12-month repeat testing window to be expanded to include all patients with secondary causes of osteoporosis, new fractures, or other verified risk factors for rapid bone loss.
The next critical step in this process is a meeting currently being arranged between representatives from Osteoporosis Canada and Ontario’s Ministry of Health to discuss the clinical gaps and the potential for policy amendments.
We invite readers to share their experiences with these testing changes in the comments below or contact your local representative to discuss access to diagnostic healthcare.

