Maintaining internal stability is a hallmark of life, but increasingly, scientists are recognizing that this stability—specifically, the balance of proteins—extends far beyond the confines of individual cells. This concept, known as extracellular proteostasis, is crucial for healthy tissue function and overall organismal well-being. Understanding how this system is organized, and what happens when it breaks down, is a rapidly evolving area of biological research with implications for a wide range of diseases, from neurodegenerative disorders to cancer.
For decades, the focus in biology was largely on intracellular proteostasis – the mechanisms cells use to create, fold, and degrade proteins. But cells don’t operate in isolation. They constantly communicate with their surroundings, releasing proteins into the extracellular space. These proteins aren’t simply waste products; they play vital roles in signaling, structural support, and immune responses. The proper regulation of these extracellular proteins, and the systems that manage them, is now understood to be just as important as what happens inside the cell. This field of extracellular proteostasis is gaining momentum as researchers uncover its complexity and significance.
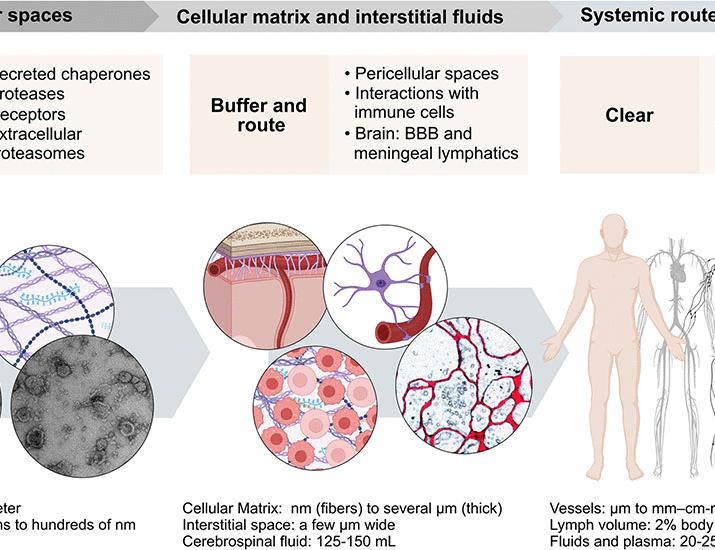
A recent review published in Science highlights the hierarchical organization of this system, describing it as operating across three interconnected tiers: the pericellular space (the immediate environment around cells), the tissue level, and the systemic level – encompassing the entire organism. This framework provides a new lens through which to view disease processes and develop potential therapeutic strategies. The authors emphasize that disruptions at any of these tiers can have cascading effects, leading to proteotoxic stress and disease.
The Three Tiers of Extracellular Proteostasis
The pericellular tier is the first line of defense. It involves proteins located in the extracellular matrix (ECM) – the network of molecules that provides structural support to tissues – and proteins directly secreted by cells. Enzymes present in this space, like proteases, break down damaged or misfolded proteins. However, this degradation isn’t indiscriminate. It’s a tightly regulated process, ensuring that only the appropriate proteins are removed. The ECM itself isn’t static; it’s constantly being remodeled, and this remodeling is likewise a key component of pericellular proteostasis.
Moving up a level, tissue-level proteostasis involves the coordinated activity of multiple cells within a specific organ or tissue. This tier relies on communication between cells, often through signaling molecules that are themselves proteins. For example, in muscle tissue, specialized cells called fibroblasts play a crucial role in removing damaged proteins from the ECM. This process is essential for maintaining muscle function and preventing age-related decline. Disruptions at the tissue level can lead to localized proteotoxic stress, contributing to conditions like fibrosis – the excessive buildup of scar tissue.
Finally, systemic proteostasis encompasses the entire organism, involving the circulation of proteins throughout the body. The liver and kidneys are key players in this tier, filtering the blood and removing damaged proteins. The immune system also plays a critical role, identifying and clearing misfolded proteins that may pose a threat. Systemic proteostasis is particularly important for maintaining overall health and preventing the spread of proteotoxic stress from one tissue to another. Conditions like chronic kidney disease can severely impair systemic proteostasis, leading to a buildup of toxins and contributing to a range of health problems.
Why Extracellular Proteostasis Matters: Disease Implications
The breakdown of extracellular proteostasis is increasingly implicated in a variety of diseases. In neurodegenerative disorders like Alzheimer’s and Parkinson’s disease, the accumulation of misfolded proteins – amyloid-beta and alpha-synuclein, respectively – is a hallmark pathology. While much of the initial research focused on the intracellular accumulation of these proteins, it’s now clear that extracellular aggregates also play a significant role in disease progression. These aggregates can trigger inflammation and disrupt neuronal communication, contributing to cognitive decline and motor dysfunction.
Cancer is another area where extracellular proteostasis is proving to be critical. Tumor cells often secrete proteins that remodel the ECM, creating an environment that promotes tumor growth and metastasis. These proteins can also suppress the immune system, allowing cancer cells to evade detection and destruction. Understanding how tumor cells manipulate extracellular proteostasis could lead to new strategies for cancer therapy.
Beyond these well-known examples, disruptions in extracellular proteostasis have also been linked to cardiovascular disease, autoimmune disorders, and even aging. As we age, the efficiency of our proteostasis systems declines, leading to a gradual accumulation of damaged proteins and contributing to age-related frailty and disease susceptibility. Research suggests that interventions aimed at boosting proteostasis – such as exercise and dietary modifications – may help to slow down the aging process and promote healthy longevity.
Future Directions and Research
The field of extracellular proteostasis is still in its early stages, and much remains to be discovered. One key challenge is developing tools to accurately measure the levels of different proteins in the extracellular space. Another is understanding how different proteostasis pathways interact with each other. Researchers are also exploring the potential of therapeutic interventions aimed at restoring extracellular proteostasis. These include developing drugs that enhance the activity of proteases, inhibit the formation of protein aggregates, or boost the immune system’s ability to clear damaged proteins.
The development of more sophisticated models – including organoids and “body-on-a-chip” systems – will be crucial for studying extracellular proteostasis in a more physiologically relevant context. These models allow researchers to recreate the complex interactions between cells and their environment, providing a more accurate picture of how proteostasis is regulated in vivo. A deeper understanding of extracellular proteostasis will pave the way for new diagnostic tools and therapies for a wide range of diseases.
The next major checkpoint in this research will likely be the results of several ongoing clinical trials investigating the potential of proteostasis-modulating therapies for Alzheimer’s disease, expected in late 2024 and 2025. ClinicalTrials.gov provides a comprehensive database of ongoing and completed clinical trials.
This emerging understanding of how our bodies maintain protein balance beyond the cell is a significant step forward in our ability to combat disease and promote healthy aging. Share your thoughts on this fascinating area of research in the comments below.