The supply chain for febuxostat 80 mg, including both the brand-name Adenuric and its generic equivalents, has largely stabilized following a period of disruption. As of early April 2026, the medication is once again available in sufficient quantities across the distribution network, providing much-needed relief for patients managing chronic gout and hyperuricemia.
The recovery of the supply chain began in mid-March 2026, allowing pharmacies and hospitals to resume standard procurement. While the majority of the logistical bottlenecks have been cleared, some healthcare providers may still encounter occasional, localized shortages depending on the specific laboratory producing the generic version.
For patients and clinicians, this return to availability necessitates a careful approach to fébuxostat 80 mg (Adenuric et génériques) : recommandations pour l’ajustement des traitements. Transitioning back to a preferred dosage or switching between generic brands requires clinical oversight to ensure that uric acid levels remain stable and that the risk of gout flares is minimized during the adjustment period.
As a physician, I have seen how volatility in medication supply can destabilize a patient’s long-term health plan. When a potent xanthine oxidase inhibitor like febuxostat becomes unavailable, patients are often shifted to alternatives or lower doses, which can lead to a rebound in serum urate levels. The current restoration of stock is a positive development, but the process of “re-stabilizing” a patient’s regimen must be handled with precision.
Navigating the Transition Back to Standard Dosing
The primary challenge following a supply shortage is not simply the availability of the pill, but the physiological response to resuming the 80 mg dose. If a patient was forced to reduce their dosage or switch to a different medication—such as allopurinol—the sudden reintroduction of a high-dose febuxostat regimen can trigger a “mobilization flare.”
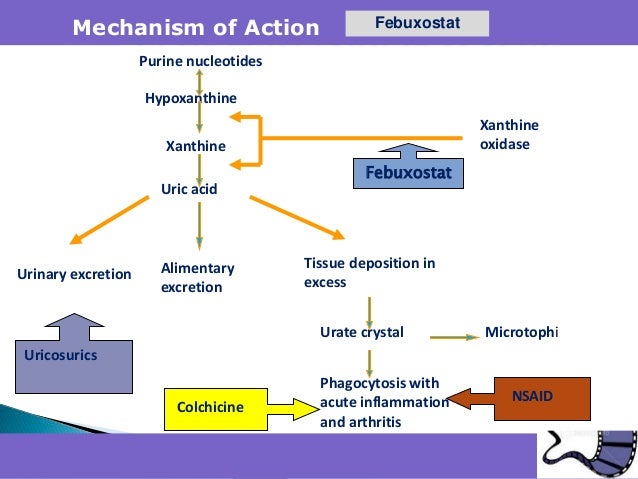
This occurs given that a rapid drop in serum uric acid levels can cause crystals to shift from the joints back into the bloodstream, paradoxically triggering an acute gout attack. To mitigate this, clinicians are advised to follow a gradual titration schedule rather than an immediate jump back to the maximum dose.
Patients should be encouraged to maintain a consistent dialogue with their prescribing physician. It is critical to monitor for signs of acute inflammation and, in some cases, utilize low-dose colchicine or corticosteroids as prophylactic coverage during the first few weeks of treatment adjustment.
Understanding the Role of Febuxostat in Gout Management
Febuxostat is specifically indicated for the long-term treatment of gout when first-line therapies, typically allopurinol, are ineffective or not tolerated. By inhibiting the enzyme xanthine oxidase, the drug reduces the production of uric acid in the body, thereby preventing the formation of the monosodium urate crystals that cause joint pain and permanent damage.
The 80 mg dose is often the target for patients who have not reached their goal serum urate level (typically below 6 mg/dL or 360 µmol/L) on lower doses. Because febuxostat is metabolized primarily by the liver, it is often a preferred choice for patients with moderate renal impairment who cannot tolerate the high doses of allopurinol required to achieve the same therapeutic effect.
For more detailed information on the pharmacological profile of these medications, the European Medicines Agency (EMA) provides comprehensive regulatory summaries on xanthine oxidase inhibitors.
Managing Generic Substitutions and Laboratory Variance
The current supply situation is characterized by “residual difficulties” among a few specific laboratories. This means that while the overall market is stocked, a patient’s local pharmacy might have one generic brand available while another is still missing. While generic versions are bioequivalent to the brand-name Adenuric, some patients report varying tolerances when switching between different generic manufacturers.
When adjusting treatments during this recovery phase, the following considerations are paramount:
- Consistency: Whenever possible, patients should attempt to stay with the same generic manufacturer to avoid unnecessary fluctuations in absorption.
- Monitoring: Serum uric acid levels should be checked 2 to 4 weeks after returning to the 80 mg dose to ensure the target level is being maintained.
- Symptom Tracking: Patients should maintain a log of any joint stiffness or redness that appears during the transition back to the full dose.
| Status Metric | Current Condition | Clinical Action |
|---|---|---|
| General Availability | Sufficient (since mid-March) | Resume standard prescribing |
| Generic Supply | Mostly stable; some lab gaps | Verify specific brand availability |
| Treatment Risk | Potential mobilization flares | Gradual titration recommended |
Clinical Implications and Patient Safety
The stability of the supply chain allows physicians to move away from “crisis prescribing”—the practice of choosing a medication based on what is on the shelf rather than what is clinically optimal. However, the transition period is a window of vulnerability.
febuxostat carries specific warnings regarding cardiovascular safety. While the 80 mg dose is effective for urate lowering, it must be used with caution in patients with a history of significant cardiovascular disease. This makes the “adjustment” phase not just about the supply of the drug, but about re-evaluating the patient’s overall cardiovascular risk profile after a gap in therapy.
For those seeking official guidelines on the management of gout and the use of urate-lowering therapies, the American College of Physicians and similar professional bodies offer evidence-based frameworks for dosing and monitoring.
Disclaimer: This article is provided for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or medication adjustment.
Healthcare providers are encouraged to continue monitoring the supply chain through official pharmaceutical portals. The next expected review of supply stability for these specific dosages will likely align with the quarterly pharmaceutical distribution reports in July 2026.
We invite readers to share their experiences with medication availability or ask questions in the comments below to help us cover these public health challenges more comprehensively.

