A new understanding of how graphene oxide – a remarkably versatile material – selectively targets and destroys bacteria while remaining harmless to human cells is emerging from research at the Korea Advanced Institute of Science and Technology (KAIST). This breakthrough in understanding graphene oxide’s antibacterial properties could pave the way for a new generation of hygienic materials and a significant reduction in our reliance on traditional antibiotics, a growing concern as antibiotic resistance continues to spread.
For years, scientists have recognized graphene oxide’s potential in biomedical applications due to its biocompatibility and ability to kill bacteria. However, the seemingly contradictory nature of these properties – how could a substance be both safe for human cells and lethal to bacteria? – remained a mystery. Researchers have now pinpointed the key: the unique surface chemistry of graphene oxide and its ability to specifically interact with a molecule found in bacterial membranes, but not in human ones.
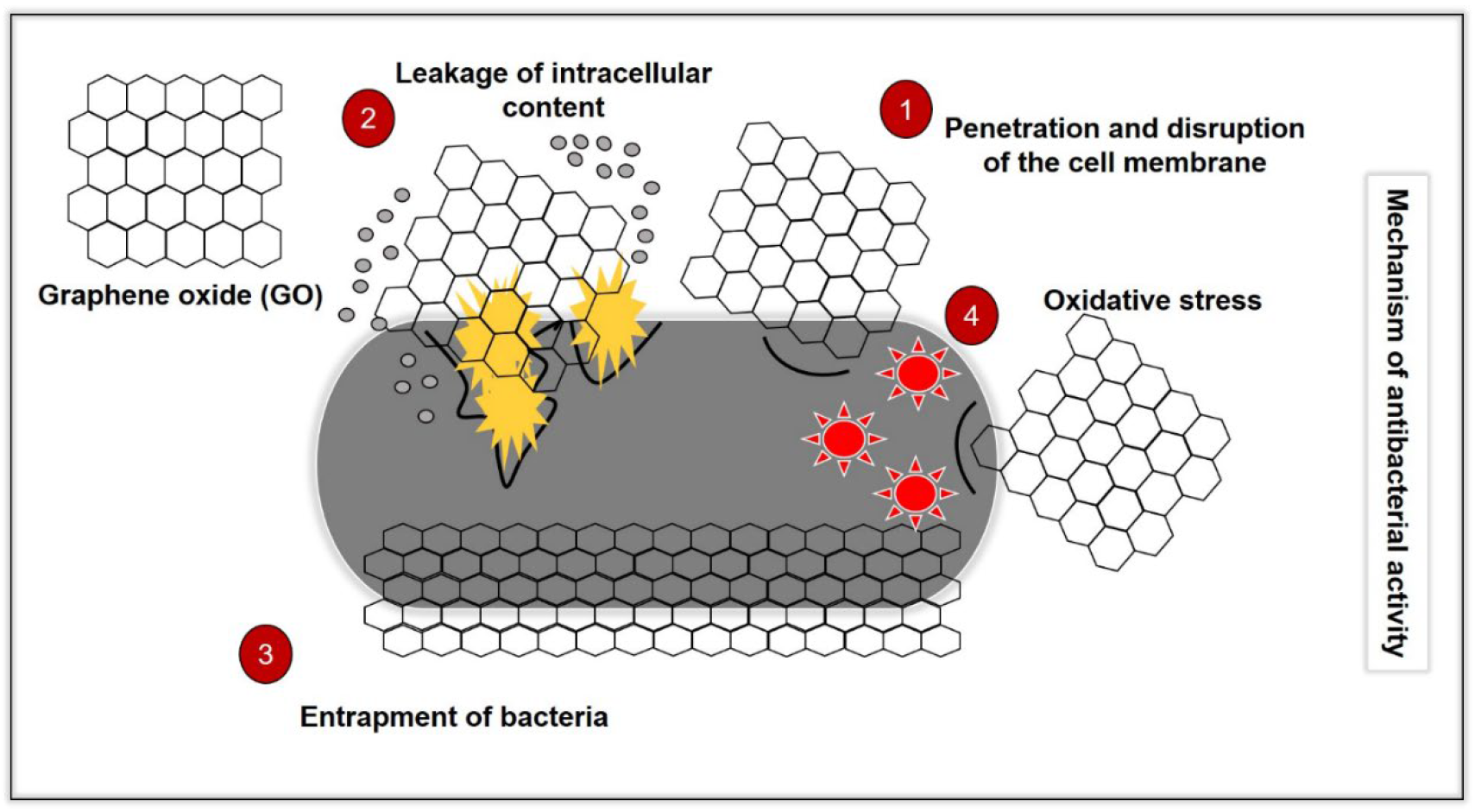
The “Lock and Target” Mechanism
The research team, led by Professor Sang Ouk Kim at KAIST, discovered that abundant oxygen-containing functional groups on the surface of graphene oxide drive a highly specific interaction with palmitoyloleoylphosphatidylglycerol (POPG). POPG is a type of phospholipid – a key building block of cell membranes – that is prominently featured in the outer layers of bacterial membranes, including those of drug-resistant strains. Mammalian cell membranes, however, contain significantly less POPG.
This difference allows graphene oxide to act like a “lock and target,” selectively binding to and destabilizing bacterial membranes. The atomically thin sheets of graphene oxide then cause large-scale disruption of the bacterial membrane, effectively killing the cell. Crucially, because human cells lack the same concentration of POPG, their membranes remain largely unaffected, explaining the material’s biocompatibility. The team utilized artificial cell models and detailed spectroscopic analyses to track these interactions at the molecular level, systematically modifying graphene oxide to understand which features drove the antibacterial activity.
From Lab to Real-World Applications
The effectiveness of graphene oxide wasn’t limited to laboratory settings. The study demonstrated its antibacterial capabilities across various formats – films, nanofibers, and powders – in animal models. In infected wound models using both mice and, importantly, pigs (whose skin is physiologically similar to human skin), graphene oxide application led to a notable suppression of bacterial growth and accelerated wound healing, with minimal inflammation or blood clotting. This preclinical data addresses inconsistencies found in previous research by directly linking performance to the defined surface chemistry and POPG-targeted mechanism.
One particularly promising application is the incorporation of graphene oxide into nanofibers. These flexible, textile-like structures effectively inhibited bacterial growth in infected wound models, promoting healing while maintaining low inflammation. Importantly, the antibacterial properties remained effective even after repeated washing, a critical requirement for reusable medical and protective equipment. This durability and unique mode of action – physically disrupting membranes rather than relying on traditional antibiotic mechanisms – distinguish graphene oxide from many conventional pharmaceuticals.
Graphene Oxide Already in Consumer Products
The technology isn’t just theoretical. Materials Creation Co., Ltd., a startup originating from KAIST faculty research, has already commercialized a graphene antibacterial toothbrush based on this patented technology. GrapheneTex, a textile material incorporating the graphene oxide mechanism, was used in the uniforms of the Taekwondo demonstration team at the 2024 Paris Olympics and is slated for leverage in functional sportswear for the 2026 Asian Games, demonstrating its readiness for high-performance athletic apparel.
Beyond consumer goods, the research suggests a broader design principle for graphene oxide-based antibacterial platforms. Because POPG and related bacterial-specific phospholipids are common across many bacterial species, including antibiotic-resistant “superbugs,” graphene oxide materials could be used as a broad-spectrum antibacterial layer in next-generation wound dressings, surgical instruments, masks, and wearable medical textiles. This could help to reduce the overuse of conventional antibiotics, a major driver of antibiotic resistance.
Addressing the Antibiotic Resistance Crisis
The World Health Organization (WHO) has identified antibiotic resistance as one of the top 10 global public health threats facing humanity. According to the WHO, antimicrobial resistance is rising globally, rendering many common infections harder to treat and increasing the risk of disease spread, severe illness, and death. New approaches, like those offered by graphene oxide, are urgently needed to combat this growing crisis.
Professor Kim emphasized that this study “is an example of scientifically uncovering why graphene can selectively kill bacteria while remaining safe for the human body.” He believes that leveraging this principle could lead to applications “beyond safe clothing without harsh chemicals to an infinite range of applications, including wearable devices and medical textile systems.”
The research provides a strong scientific foundation for graphene oxide as a versatile, long-term antibacterial material, aligning with both clinical needs and practical performance in real-world settings. The next step involves further clinical trials and scaling up production to meet potential demand. Researchers are also exploring ways to optimize the graphene oxide surface chemistry to enhance its antibacterial activity and broaden its spectrum of effectiveness.
Have thoughts on this exciting development? Share your comments below, and please share this article with your network.