On April 9, 1982, the scientific community encountered a discovery that threatened to upend the most fundamental rule of molecular biology. Dr. Stanley Prusiner, a researcher at the University of California, San Francisco, published a paper in the journal Science proposing that a protein, devoid of any genetic material, could act as an infectious agent. This hypothesis introduced the world to transmissible proteins, which Prusiner termed “prions.”
For decades, the “central dogma” of biology held that genetic information flows in one direction: from DNA to RNA to protein. In this framework, proteins were the end products—the workers of the cell—not the architects of disease. The idea that a protein could replicate itself and cause a fatal brain disease without the help of DNA or RNA was not just revolutionary; to many of his peers, it was biologically impossible.
Prusiner’s operate focused on a group of relentless, degenerative nerve diseases known as spongiform encephalopathies. These conditions are characterized by a “spongiform” appearance under the microscope, where the brain tissue becomes riddled with holes, resembling a sponge. Among these were scrapie in sheep and goats, kuru in humans, and the devastating Creutzfeldt-Jakob disease (CJD).
The catalyst for Prusiner’s obsession was a personal encounter early in his career. Even as beginning a residency in neurology, he witnessed a patient die rapidly from CJD. The disease destroyed the woman’s brain in just two months, yet her body remained otherwise unaffected, and she showed no fever or signs of an immune response. “I was most impressed by a disease process that could kill my patient in two months by destroying her brain while her body remained unaffected by this process,” Prusiner later recalled in a speech regarding his discovery.
The Mystery of the ‘Slow Virus’
Before the discovery of prions, the medical community struggled to explain how these diseases spread. In the 1950s and 60s, researchers used the term “slow virus” to describe agents that took years to manifest symptoms. This was applied to scrapie in sheep and kuru—a disease that affected the Fore tribe in Papua New Guinea, seemingly transmitted through the ritual consumption of deceased relatives’ brain tissue.

The puzzle deepened with Creutzfeldt-Jakob disease. While CJD could be transmitted to chimpanzees via infected brain tissue, the disease also appeared to run in families. This created a paradox: how could a pathogen be both infectious (like a virus) and heritable (like a genetic mutation)?
Prusiner found a critical clue in the work of radiobiologist Tikvah Alper. Alper discovered that scrapie remained transmissible even after infected tissue was irradiated with ultraviolet light—a process that typically destroys DNA and RNA. If the “virus” could survive the destruction of its genetic blueprint, perhaps the blueprint wasn’t there to commence with.
From Mice to Hamsters: The Path to Proof
To isolate the infectious agent, Prusiner began experimenting with animal models. He initially studied scrapie in mice, but found the incubation period too long, often taking one to two years for symptoms to appear. He pivoted to hamsters, which developed the disease within approximately 70 days, allowing for a much faster cycle of observation and experimentation.
Prusiner’s systematic approach led him to a startling conclusion: the infectious agent was a protein. In his 1982 study, he presented six distinct lines of evidence showing that a protein was required for infectivity. Crucially, when the protein structure was broken down, the transmission of scrapie was short-circuited. He found no evidence of nucleic acids in the samples, leading him to propose that the protein could “code for its own biosynthesis.”
A Paradigm Shift in Molecular Biology
The scientific community did not embrace the prion hypothesis immediately. The idea that a protein could act as an infectious agent without DNA was seen as a violation of biological law. However, over the next 15 years, the evidence mounted. Researchers discovered that prions are not foreign proteins, but rather misfolded versions of a normal protein already present in the body.
When a misfolded prion comes into contact with a healthy version of the same protein, it “converts” the healthy protein into the pathological shape. This creates a domino effect, leading to the accumulation of protein aggregates that resist degradation and eventually destroy neurons, leaving the brain riddled with the characteristic holes of spongiform encephalopathy.
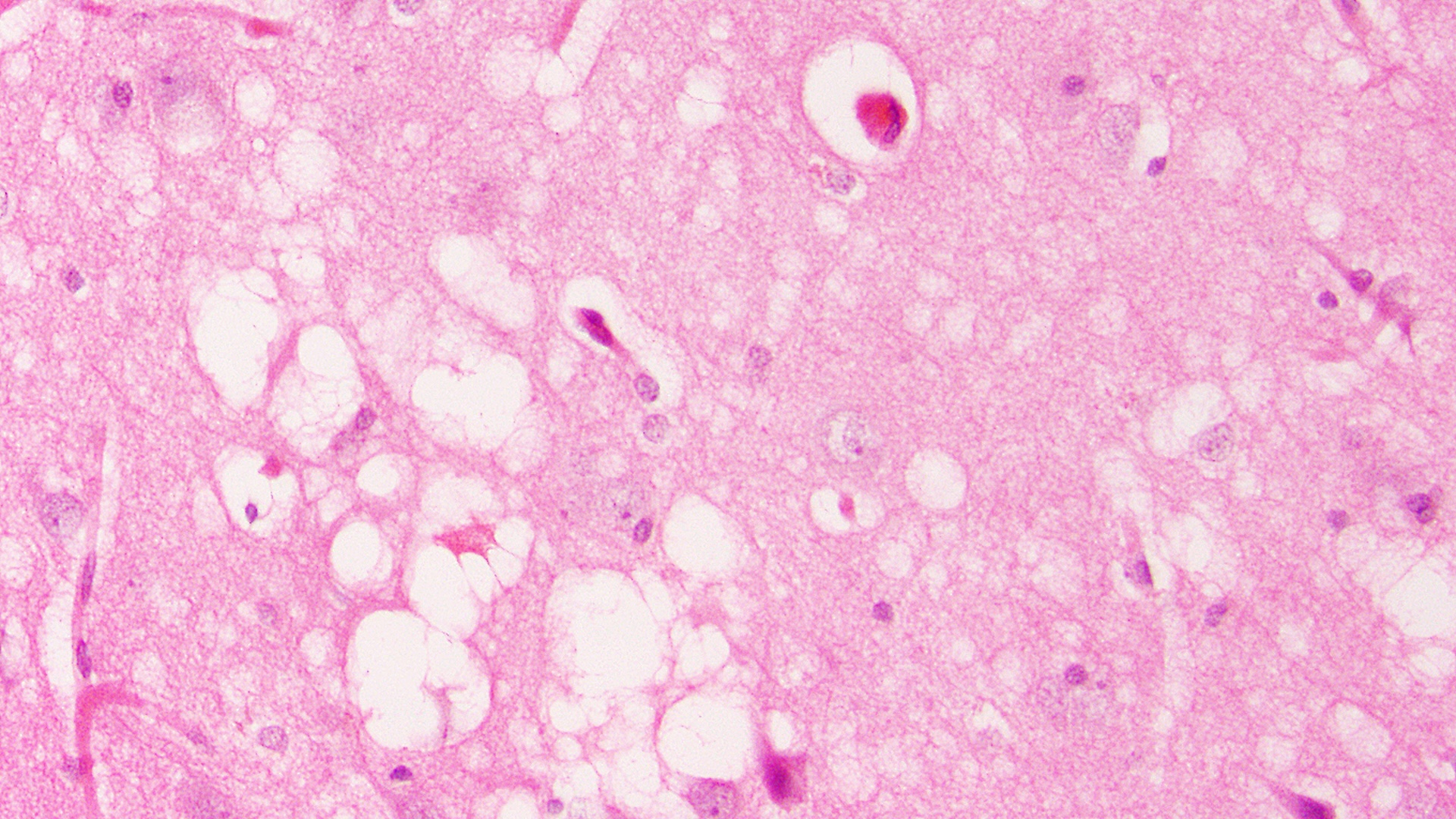
Impact and Validation
The validation of Prusiner’s theory came with devastating real-world consequences. In the early 2000s, the United Kingdom was struck by an epidemic of “mad cow disease,” known scientifically as bovine spongiform encephalopathy (BSE). It was later determined that the disease spread when cows were fed brain tissue from other infected cows.
Humans who consumed beef from cows with BSE developed a version of CJD known as variant CJD (vCJD). This tragedy underscored the terrifying stability of prions; they are resistant to standard sterilization methods that would easily kill bacteria or viruses.
For his courage in challenging the established dogma and his rigorous proof of the existence of prions, Dr. Stanley Prusiner was awarded the Nobel Prize in Physiology or Medicine in 1997.
| Period/Date | Key Event | Significance |
|---|---|---|
| 1950s-60s | “Slow Virus” Hypothesis | Used to describe scrapie and kuru before the agent was known. |
| April 9, 1982 | Prusiner’s Science Paper | First proposal that proteins alone could cause infectious disease. |
| 1980s-90s | Protein Folding Research | Discovery of how prions convert healthy proteins into pathological forms. |
| 1997 | Nobel Prize Award | Official scientific recognition of the prion hypothesis. |
| Early 2000s | UK Mad Cow Epidemic | Real-world validation of prion transmission from animals to humans. |
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Please consult a healthcare professional for questions regarding neurological health.
Today, the study of prions extends beyond rare infectious diseases. Researchers are investigating whether similar “prion-like” protein misfolding plays a role in more common neurodegenerative conditions, such as Alzheimer’s and Parkinson’s diseases. The next major milestone in this field remains the development of a viable therapeutic to stop or reverse the misfolding process, a challenge that continues to occupy the world’s leading neurologists.
We invite you to share your thoughts on this scientific milestone in the comments below.