For four decades, the Trypanosoma brucei parasite has played a high-stakes game of hide-and-seek with the human immune system, utilizing a biological disguise that baffled researchers for nearly half a century. A team from the University of York has now solved this long-standing sleeping sickness parasite mystery, identifying the specific cellular mechanism the pathogen uses to maintain its invisibility.
The discovery centers on a specialized protein that acts as a “molecular shredder,” allowing the parasite to prioritize the production of its protective outer layer while aggressively destroying other genetic instructions. This efficiency prevents the organism from overloading its own cellular machinery, ensuring it can continuously evolve its appearance to stay one step ahead of the host’s defenses.
Published in Nature Microbiology, the study reveals how the parasite manages a precarious balance of protein production. By identifying this regulator, scientists have opened a new door for developing treatments that could potentially strip the parasite of its disguise, making it vulnerable to the body’s natural immune response.
The Biology of Invisibility
The African Trypanosoma brucei parasite is the causative agent of sleeping sickness, a disease known clinically as human African trypanosomiasis. Its primary survival strategy is the use of a “molecular cloak” composed of variant surface glycoproteins (VSG). This outer coating is not static; the parasite constantly renews and alters the VSG proteins, effectively changing its “face” before the immune system can develop the antibodies necessary to recognize and destroy it.

While scientists have long known about the existence of this cloak, the manufacturing process remained an enigma. Specifically, researchers could not determine how the parasite kept the production of VSG at such an intense level without exhausting its energy or interfering with the production of other essential proteins needed for survival, such as those used to steal nutrients from the host.
The University of York team discovered that the answer lies in a protein they named ESB2. This protein sits at the end of the cell’s production line, acting as a selective filter for RNA molecules—the instructions that tell the cell which proteins to build.
Inside the Molecular Shredder
ESB2 functions by selectively destroying RNA. It allows the instructions for the VSG “cloak” to pass through untouched, while chopping up the instructions for other proteins. This ensures that the parasite does not waste energy on unnecessary proteins, focusing its entire metabolic effort on the disguise that keeps it alive.
“Imagine a molecular shredder stationed right inside the genetic print room, selectively redacting specific pages of a manual as they are being printed,” says microbiologist Joana Correia Faria, the senior author of the study. “It is a remarkably efficient way to downregulate genes at the source.”
To uncover this mechanism, the researchers used mass spectrometry to tag and identify proteins within the parasite’s cells. Once ESB2 was identified, the team used genetic editing to remove the protein from T. Brucei. Without the “shredder” in place, the production of helper proteins shot up to match the levels of the VSG proteins, confirming that ESB2’s primary role was to suppress non-essential protein production.

The Human Cost of Sleeping Sickness
The biological ingenuity of T. Brucei has devastating consequences for those infected. The disease is transmitted through the bite of the tsetse fly, primarily in sub-Saharan Africa. Once in the bloodstream, the parasite eventually crosses the blood-brain barrier, attacking the central nervous system.

The resulting symptoms often mirror the disease’s name, leading to disrupted sleep cycles, profound confusion, and eventually comas. If left untreated, the infection is almost always fatal. While global efforts have reduced the number of cases, the disease still affects hundreds of people annually, often in remote areas where medical access is limited.
Current treatments exist but are often fraught with challenges. Some are difficult to administer in rural settings, while others carry the risk of severe side effects or may not be effective against all strains of the parasite. This makes the discovery of a new biological vulnerability like ESB2 particularly significant.
A New Path for Treatment
The identification of ESB2 suggests a fundamental shift in how scientists might approach treating parasitic infections. Rather than focusing solely on how an organism issues genetic instructions, the University of York team suggests that survival may depend more on how those instructions are destroyed.
By developing drugs that disrupt or inhibit the activity of ESB2, researchers believe they could potentially “break” the parasite’s ability to manage its protein production. This could lead to two possible outcomes: the parasite could be killed by its own metabolic overload, or it could be forced to produce proteins that make it visible to the host’s immune system, allowing the body to clear the infection naturally.
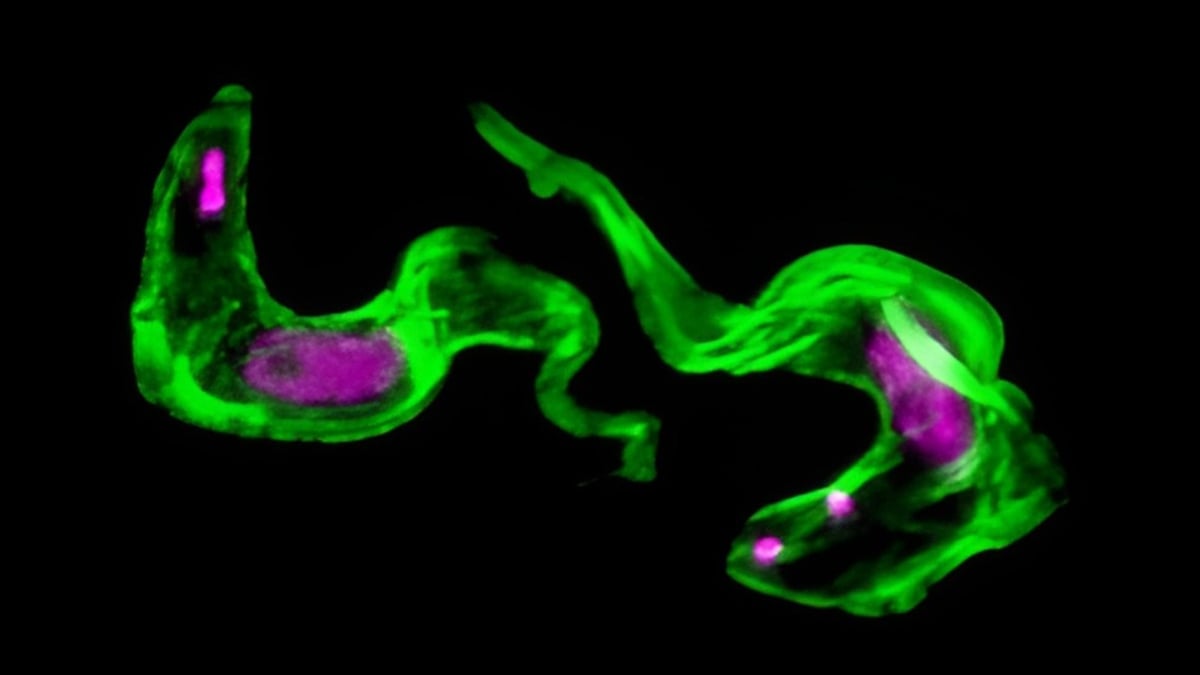
Microbiologist Lianne Lansink, the study’s first author, noted that seeing the molecular shredder localized under a microscope was a pivotal moment for the team, signaling that they had found a “special” mechanism that had remained hidden for decades.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Please consult a healthcare professional for diagnosis and treatment of any medical condition.
The research team now plans to investigate exactly how ESB2 identifies which RNA molecules to shred and which to spare. They will also examine the other proteins that ESB2 interacts with to determine if You’ll see additional vulnerabilities in the parasite’s cellular machinery. These next steps will be critical in determining if ESB2 can be successfully targeted by pharmaceutical interventions.
Do you think targeting RNA degradation is the future of anti-parasitic medicine? Share your thoughts in the comments below.