For millions of families, the journey toward a dementia diagnosis is often a grueling process of elimination, marked by vague symptoms and a long wait for definitive answers. However, researchers at the Yale School of Medicine are developing a neuroimaging technique that could significantly speed up the detection of neurodegenerative diseases, potentially identifying markers of cognitive decline long before clinical symptoms become severe.
The challenge with diagnosing dementia, particularly Alzheimer’s disease, is that by the time a patient exhibits noticeable memory loss or behavioral changes, the brain has often undergone extensive damage. The Yale team is focusing on the “preclinical” stage—the window where the brain is changing, but the person still functions normally. By utilizing advanced imaging and computational analysis, the researchers aim to pinpoint the exact moment a brain begins to deviate from healthy aging toward a pathological state.
This shift toward earlier detection is not merely about a faster label; it is about the critical window for intervention. As new disease-modifying therapies emerge, the ability to identify candidates for treatment in the earliest stages of the disease could fundamentally change the trajectory of patient care and long-term outcomes.
The Role of Advanced Neuroimaging in Early Detection
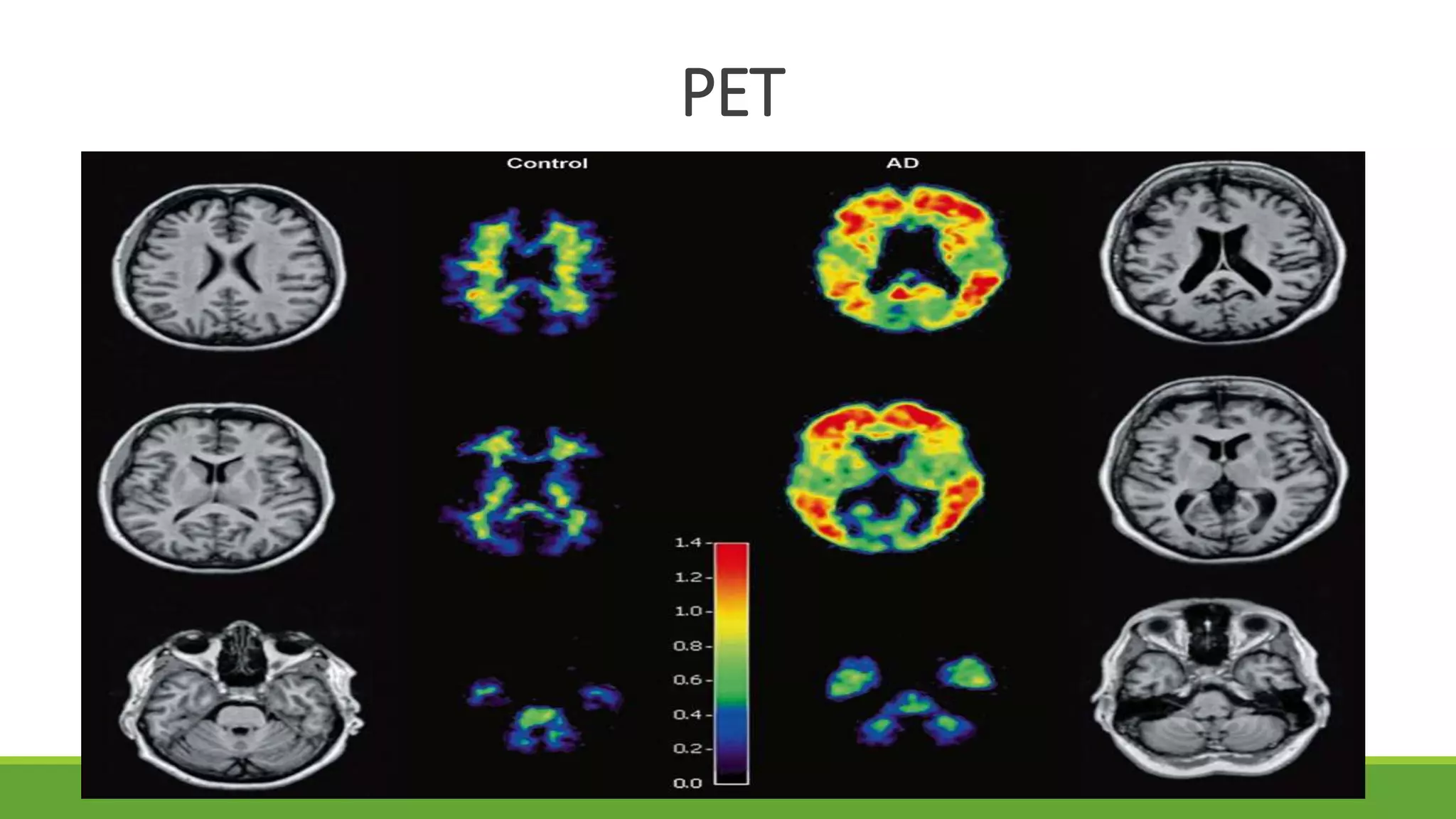
Traditional imaging, such as standard MRI scans, often looks for “atrophy”—the shrinking of brain tissue. Even as effective, atrophy is a late-stage marker. The Yale research focuses on more subtle changes in brain structure and connectivity that precede visible shrinkage. By applying sophisticated algorithms to neuroimaging data, the team can detect “hidden” patterns of degeneration that are invisible to the naked eye of a radiologist.
This approach leverages the concept of biological signatures. Just as a blood test can reveal a predisposition to diabetes before a patient feels ill, these neuroimaging techniques look for the structural “fingerprints” of dementia. This allows clinicians to move away from a diagnosis based primarily on behavioral observation and toward one based on objective biological evidence.
The integration of artificial intelligence (AI) plays a pivotal role in this process. The researchers use machine learning to compare an individual’s brain scan against vast databases of both healthy and diseased brains. This allows the system to identify minute deviations in the thickness of the cerebral cortex or the integrity of white matter tracts, which are often the first areas affected by protein build-up in the brain.
Bridging the Gap Between Research and Clinic
While the technology is promising, transitioning from a laboratory setting to a standard clinical environment requires overcoming several hurdles. One primary concern is the accessibility of high-resolution imaging. Not every community clinic has the specialized equipment or the computational power required to run these advanced analyses.
The Yale team is working to streamline these tools so they can be integrated into existing healthcare workflows. The goal is to create a “screening” layer where a standard scan can be flagged by an algorithm for further, more detailed review if certain markers are present. This would prevent the healthcare system from being overwhelmed while ensuring that high-risk patients are fast-tracked for specialized care.
The impact of this speed in detection is most evident when considering the current diagnostic timeline. Many patients spend years visiting different specialists—primary care physicians, neurologists, and neuropsychologists—before receiving a definitive diagnosis. A validated neuroimaging biomarker could reduce this “diagnostic odyssey,” providing families with clarity and a roadmap for the future much sooner.
Who Benefits From Earlier Diagnosis?
The primary stakeholders in this technological shift are the patients and their caregivers, but the implications extend to the broader public health infrastructure. Early detection allows for a proactive approach to healthcare, shifting the focus from palliative care to preventative and modifying strategies.

- Patients: Earlier diagnosis provides the opportunity to participate in clinical trials and access new medications that are most effective when administered early.
- Caregivers: Families can build informed decisions about legal, financial, and living arrangements while the patient still possesses the cognitive capacity to participate in those conversations.
- Healthcare Systems: More accurate early screening can reduce the cost of unnecessary tests and allow for more efficient allocation of specialized memory care resources.
However, the ability to detect a disease before symptoms appear also brings ethical complexities. The medical community must balance the benefit of early intervention with the psychological impact of knowing one has a progressive disease before they feel “sick.” This necessitates a multidisciplinary approach involving not just neurologists, but genetic counselors and mental health professionals.
Comparing Traditional vs. Advanced Detection
To understand the leap in technology, it is helpful to compare the traditional diagnostic path with the proposed neuroimaging-led approach.
| Feature | Traditional Diagnosis | Advanced Neuroimaging |
|---|---|---|
| Primary Trigger | Observable cognitive decline | Biological markers/AI analysis |
| Timing | Symptomatic stage | Preclinical/Early stage |
| Key Evidence | Cognitive tests & brain atrophy | Structural connectivity patterns |
| Diagnostic Speed | Often months or years | Potentially rapid once scanned |
The Path Toward Clinical Implementation
The next steps for the Yale researchers involve validating these imaging markers across larger, more diverse populations. For a diagnostic tool to be reliable, it must perform consistently across different ages, ethnicities, and comorbidities. A brain that shows signs of aging due to chronic hypertension, for example, must be distinguishable from a brain showing early signs of Alzheimer’s.
the research team is exploring the correlation between these imaging findings and other biomarkers, such as those found in cerebrospinal fluid (CSF) or blood-based tests. The ultimate goal is a “multimodal” diagnostic approach—combining a simple blood test with an advanced neuroimaging scan to achieve near-certainty in a diagnosis.
As the National Institute on Aging continues to fund research into the biological definitions of dementia, the work at Yale aligns with a global shift toward treating dementia as a biological disease that begins decades before the first forgotten name or lost key.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
The researchers are now moving toward larger longitudinal studies to track how these early imaging markers evolve over time. The next major checkpoint will be the publication of peer-reviewed data demonstrating the predictive accuracy of these tools in a real-world clinical cohort, which will be essential for seeking regulatory approval for clinical use.
We invite you to share your thoughts on the balance between early detection and psychological readiness in the comments below.