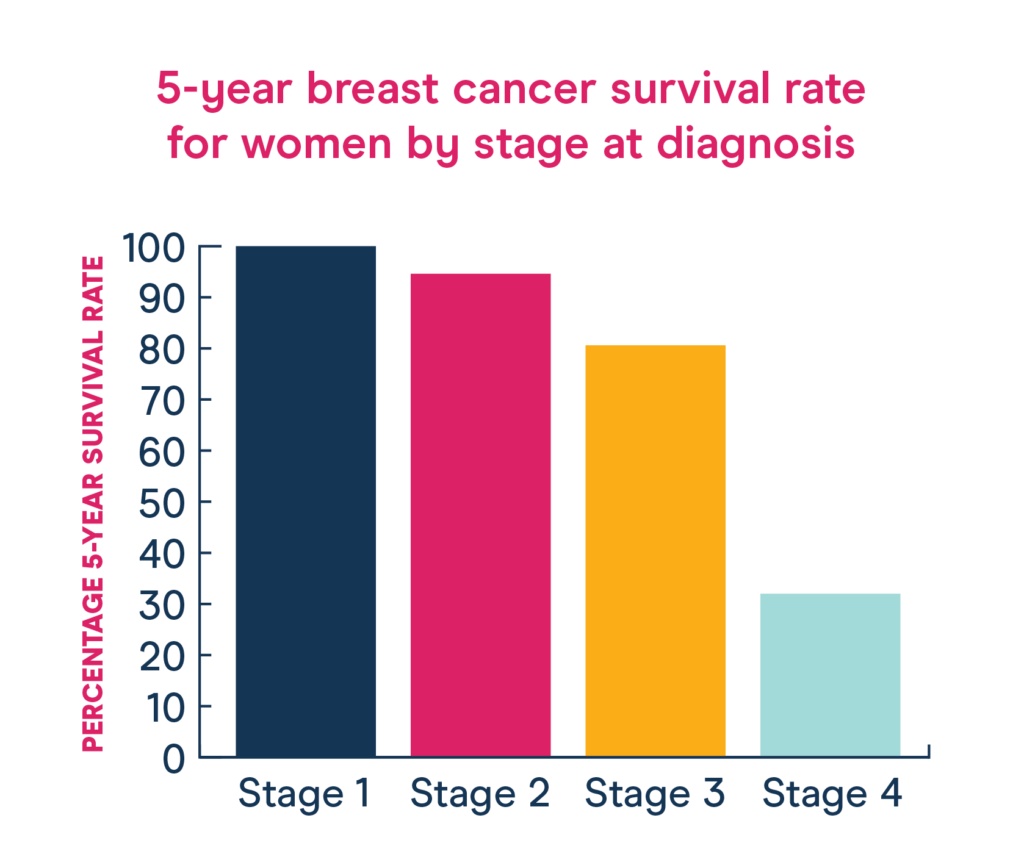
For oncology patients, the five-year prognosis is more than a statistic. it is a roadmap for treatment, recovery and emotional planning. Historically, determining this outlook has relied on the expertise of pathologists analyzing tissue slides under microscopes—a process that, while rigorous, is subject to human variability. However, the integration of transfer learning for breast cancer prognosis is fundamentally changing how clinicians predict long-term patient outcomes.
By leveraging artificial intelligence models that have already “learned” to recognize shapes and patterns from millions of general images, researchers are now able to identify subtle cellular markers of malignancy that often escape the human eye. This approach allows for a more precise determination of whether a cancer is likely to recur or remain dormant over a five-year period, potentially sparing patients from unnecessary aggressive chemotherapy or identifying those who need more intensive intervention.
This shift toward AI-driven diagnostics is not about replacing the pathologist, but rather providing them with a high-resolution digital second opinion. As a former software engineer, I find the elegance of this transition particularly striking: we are taking the “intelligence” gained from identifying everyday objects—like dogs or cars—and refining it to detect the microscopic architecture of a tumor.
The Mechanics of Transfer Learning in Oncology
In traditional deep learning, a model starts as a blank slate. To train it to recognize breast cancer cells, a developer would need hundreds of thousands of labeled medical images, which are notoriously difficult to acquire due to patient privacy laws and the high cost of expert annotation. This “data scarcity” has long been a bottleneck in medical AI.
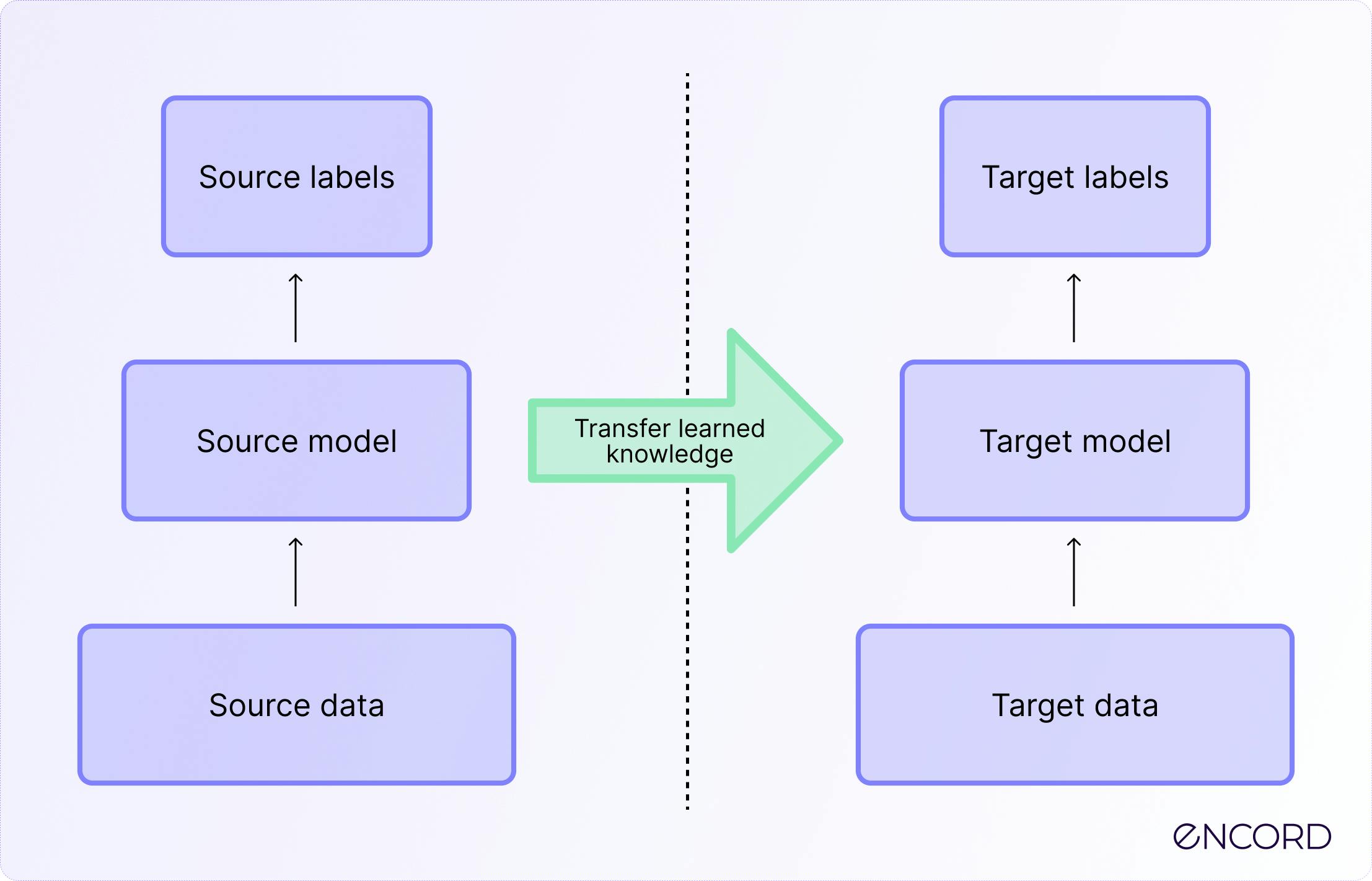
Transfer learning solves this by using a pre-trained model, typically one trained on the ImageNet dataset, which contains over 14 million images across thousands of categories. While a picture of a golden retriever has nothing in common with a biopsy slide, the early layers of the neural network learn universal features: edges, curves, textures, and gradients. These basic visual building blocks are the same whether the AI is looking at a landscape or a lymphocyte.
The process involves “freezing” these early layers and adding new, specialized layers at the end of the network. These final layers are then “fine-tuned” using a smaller, high-quality dataset of breast cancer histopathology slides. The result is a model that possesses the general visual intelligence of a massive dataset but the specific diagnostic precision of an oncologist.
Comparing Learning Approaches in Medical Imaging
The efficiency gain when moving from scratch-built models to transfer learning is significant, particularly regarding the amount of data required to reach a stable predictive accuracy.
| Feature | Traditional Deep Learning | Transfer Learning |
|---|---|---|
| Data Requirement | Massive labeled medical datasets | Small to medium specialized datasets |
| Training Time | Weeks or months of compute | Hours or days (fine-tuning) |
| Initial State | Randomly initialized weights | Pre-trained on general images |
| Risk of Overfitting | High with small datasets | Lower due to pre-existing feature knowledge |
Predicting the Five-Year Window
The “five-year” mark is a critical benchmark in oncology. In many breast cancer cases, if a patient remains cancer-free for five years, the likelihood of recurrence drops significantly. Predicting this outcome requires the AI to look beyond the mere presence of cancer and instead analyze the “tumor microenvironment”—the way cancer cells interact with surrounding healthy tissue, blood vessels, and immune cells.
Research published through platforms like PubMed suggests that deep learning models can identify “morphological signatures” associated with poor prognosis that are too complex for human categorization. These might include specific patterns of nuclear pleomorphism (variation in nucleus size and shape) or the density of tumor-infiltrating lymphocytes.
When these AI models are applied to the National Cancer Institute’s guidelines and patient data, they can help stratify patients into low-risk and high-risk groups with greater accuracy than traditional TNM (Tumor, Node, Metastasis) staging alone. This stratification is essential for personalized medicine, ensuring that treatment is scaled to the actual risk of the individual.
The Challenges of the ‘Black Box’
Despite the promise, the primary hurdle for transfer learning for breast cancer prognosis is interpretability. In medicine, “the AI said so” is not an acceptable justification for a surgical procedure or a toxic drug regimen. Neural networks are often criticized as “black boxes” because it is difficult to see exactly which pixels triggered a high-risk prediction.
To combat this, researchers are implementing “heat maps” or attention maps. These tools highlight the specific areas of a biopsy slide that the AI focused on to reach its conclusion. When a pathologist sees that the AI is flagging the same invasive fronts they would have noticed, trust in the system grows. When the AI flags something the pathologist missed, it prompts a deeper human review, creating a symbiotic loop of verification.
there is the issue of “dataset bias.” If a model is trained primarily on images from one demographic or one specific type of scanning equipment, its accuracy may drop when applied to a different population. Ensuring that these models are trained on diverse, global datasets is a current priority for the medical AI community.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
The next major milestone for this technology will be the transition from retrospective studies—where AI is tested on old data—to prospective clinical trials. These trials will determine if AI-guided prognosis actually improves patient survival rates in real-time clinical settings. As these tools move toward FDA clearance and integration into digital pathology workflows, the goal remains clear: turning data into time for the people who need it most.
Do you believe AI will eventually lead the way in cancer diagnosis, or should it always remain a secondary tool? Share your thoughts in the comments below.