Early detection remains the most critical factor in surviving oral cancer, yet for many patients, the first red flag is often missed during routine dental visits. Due to the fact that many early-stage lesions are nearly indistinguishable from benign sores to the naked eye, clinicians have long relied on a combination of visual inspection and clinical intuition. Now, a new integration of mobile technology and artificial intelligence is attempting to close that diagnostic gap.
Researchers at Rice University have developed a specialized un outil d’imagerie sur smartphone known as mDOC (mobile detection of oral cancer). The system is designed to act as a digital second opinion for dentists, utilizing a combination of advanced light physics and machine learning to identify tissue alterations that are invisible under standard clinic lighting. By shifting the screening process from a purely subjective visual exam to a data-driven analysis, the tool aims to increase the reliability of triage in general practice.
The urgency for such a tool is underscored by sobering statistics. According to the American Cancer Society, there were 12,770 deaths linked to oral cancers in the United States in 2025. A significant portion of these fatalities could potentially be avoided if the disease were caught in its earliest, most treatable stages, rather than after it has progressed to a more aggressive state.
The Science of Autofluorescence and AI
The mDOC system does not rely on a standard camera alone. Instead, it employs a dual-imaging approach. First, it captures traditional white-light images. Second, it uses autofluorescence imaging at a wavelength of 405 nm. This specific frequency of light causes certain biological tissues to glow; when those tissues become cancerous or precancerous, they lose this natural fluorescence, creating “dark spots” or anomalies that the human eye cannot see.
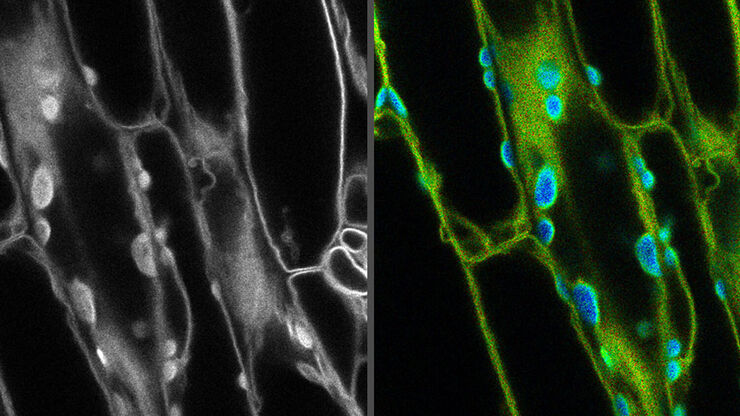
To move from a simple image to a clinical recommendation, the system integrates the visual data with the patient’s specific risk profile. This includes variables such as age, smoking history, and the exact anatomical location of the lesion. This combined dataset is then processed by a machine learning algorithm designed to recognize patterns associated with malignancy.
The practical application of this technology is designed for speed. In a study published in Biophotonics Discovery in 2025, the data acquisition process took approximately 3.5 minutes. This brief window allows the tool to be integrated into a routine dental cleaning or check-up without significantly disrupting the clinic’s workflow.
Technical Specifications of the mDOC System
| Feature | Specification / Detail |
|---|---|
| Imaging Modes | White light + 405 nm Autofluorescence |
| Analysis Method | Machine Learning (AI) Algorithm |
| Key Input Data | Images, Age, Smoking Status, Lesion Site |
| Processing Time | ~3.5 minutes for data acquisition |
| Clinical Goal | Triage and specialist referral recommendation |
Addressing the ‘Under-Served’ Gap in Public Health
One of the primary drivers behind the development of this smartphone-based tool is the disparity in healthcare access. High-end diagnostic equipment is often concentrated in large urban hospitals or specialized oncology centers, leaving patients in rural or under-funded areas with limited options. Because almost every dental office possesses a smartphone, the mDOC system effectively decentralizes high-tech screening.
The tool is not intended to replace the biopsy—the gold standard for cancer diagnosis—but rather to act as a more accurate “filter.” In a trial involving 50 patients in a dental setting, the focus was on improving the reliability of sorting lesions, particularly those with low prevalence where a dentist might be less likely to suspect cancer based on visual cues alone.
By providing a recommendation for referral, the AI helps practitioners decide which patients need an urgent appointment with an oral surgeon and which can be monitored. This reduces the burden on specialists although ensuring that high-risk patients are not sent home with a “wait and see” instruction that could prove fatal.
Clinical Implications and Next Steps
As a physician, I recognize that the integration of AI into primary care is often met with caution. The challenge for mDOC will be maintaining a high level of specificity—minimizing false positives that could lead to unnecessary patient anxiety and costly, invasive biopsies. However, the potential to catch a squamous cell carcinoma in its earliest stage far outweighs the inconvenience of a cautious referral.
The transition from a research study of 50 patients to a global clinical tool will require larger, multi-center trials to validate the algorithm across diverse populations and different smartphone hardware. The goal is to move toward a standardized “digital oral exam” that becomes as common as checking blood pressure.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
The next phase for this technology involves further optimization of the machine learning models and potential regulatory filings to move the device from a research prototype to a certified medical tool. Updates on these clinical trials will be essential for determining when the tool may be available for widespread use in dental practices.
We invite you to share your thoughts on the role of AI in routine health screenings in the comments below.

