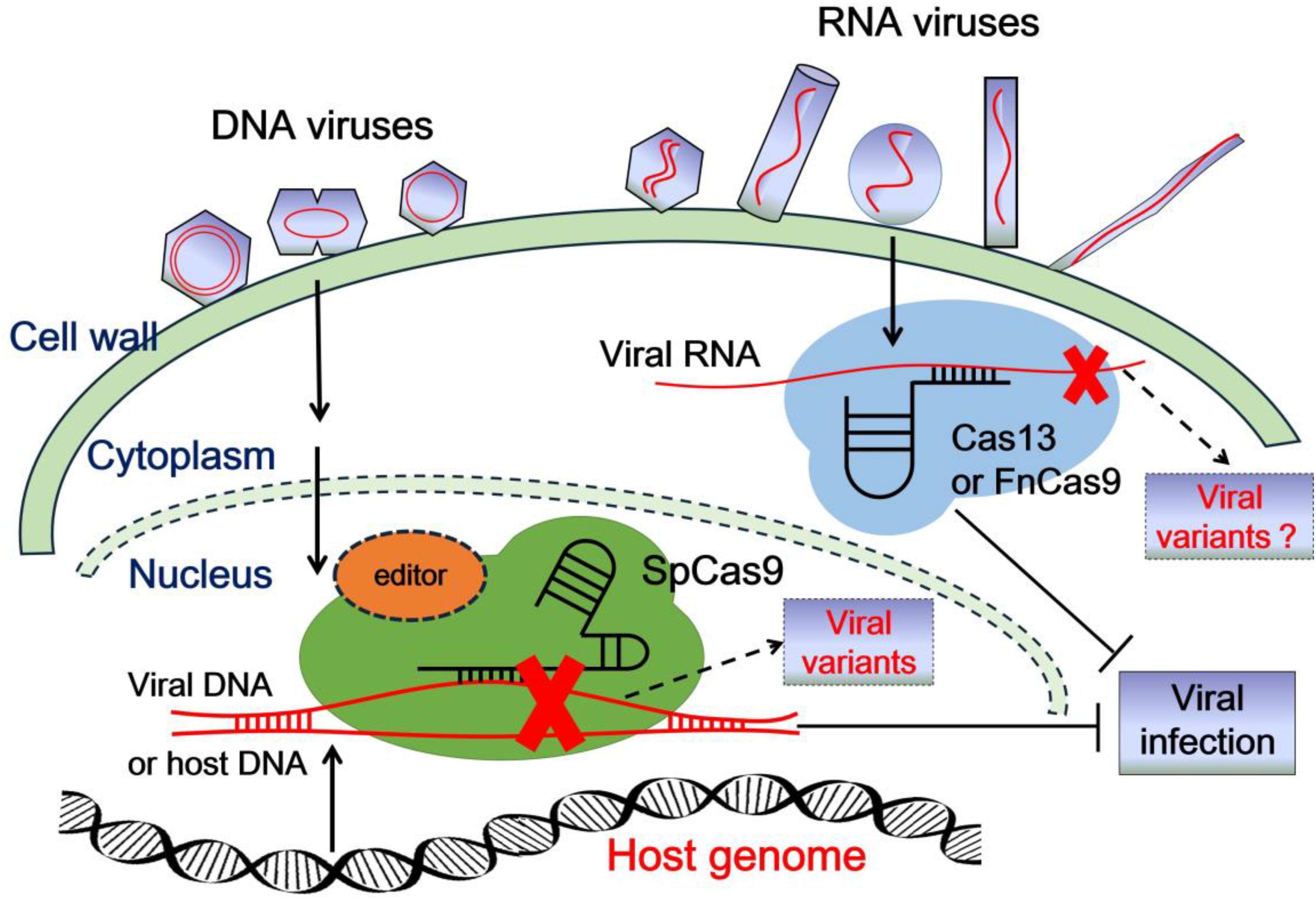
Scientists at the Hong Kong University of Science and Technology (HKUST) have developed a new method for genetic intervention that replaces the traditional RNA guide with a DNA counterpart. This shift toward DNA-guided CRISPR for RNA editing offers a more stable and precise way to identify and neutralize viral pathogens without permanently modifying a patient’s underlying genomic structure.
While CRISPR technology is most famous for its ability to “cut and paste” DNA, the focus has increasingly shifted toward RNA. Because RNA acts as the messenger between DNA and proteins, editing it allows for temporary, reversible changes. This makes it a safer alternative for treating diseases where a permanent genetic change would be too risky or unnecessary. However, the traditional RNA guides used to direct these molecular scissors are notoriously fragile and prone to degradation.
The HKUST research team addressed this instability by engineering a system that uses DNA to guide the CRISPR protein to its RNA target. By leveraging the inherent durability of DNA, the researchers have created a tool that is not only more robust but also easier to synthesize and deploy for rapid virus detection and potential antiviral therapies.
Solving the stability problem in RNA targeting
In standard CRISPR-Cas systems, a piece of RNA (the guide RNA) is used to recognize a matching sequence of genetic material. When targeting RNA viruses or messenger RNA, the guide itself is subject to the same enzymatic breakdown as the target. This instability often limits the efficiency of the tool, especially in complex biological environments.
The move to a DNA-guided system changes the fundamental chemistry of the search. DNA is significantly more stable than RNA, meaning the guide can persist longer in the cell or a diagnostic sample, increasing the likelihood that it will find and bind to the target viral RNA. This architectural shift allows the system to maintain high specificity while improving the overall reliability of the detection process.
From a manufacturing perspective, DNA guides are also simpler to produce at scale. Synthetic DNA is cheaper and more stable to transport than RNA, which often requires stringent cold-chain logistics. For clinicians and researchers, this means a more accessible tool for identifying pathogens in real-time.
Applications in rapid virus detection
One of the most immediate applications of this technology is in the realm of diagnostics. The DNA-guided CRISPR tool can be programmed to recognize the unique genetic signatures of various RNA viruses, including those responsible for influenza and coronaviruses. When the DNA guide finds its matching RNA target, the CRISPR protein triggers a signal that can be read as a positive test result.
This method provides a bridge between the speed of rapid antigen tests and the accuracy of PCR (polymerase chain reaction) tests. Because the DNA-guided system is highly sensitive, it can detect viral loads that might be missed by simpler tests, while avoiding the lengthy laboratory processing times associated with traditional genomic sequencing.
The precision of this tool also reduces “off-target” effects—instances where the CRISPR system accidentally binds to a non-target sequence. By optimizing the DNA guide, the researchers have increased the fidelity of the system, ensuring that the tool only reacts when the specific pathogen is present.
Comparison of CRISPR guiding mechanisms
| Feature | RNA-Guided CRISPR | DNA-Guided CRISPR |
|---|---|---|
| Guide Stability | Low (prone to degradation) | High (chemically robust) |
| Synthesis Cost | Higher | Lower |
| Target Material | DNA or RNA | Primarily RNA |
| Primary Use Case | Genome editing / Gene knockout | Diagnostics / RNA modulation |
A new path for antiviral therapeutics
Beyond diagnostics, the ability to precisely target RNA opens new doors for treating viral infections. Most traditional antivirals work by interfering with a virus’s ability to replicate, but they often struggle to keep up with rapidly mutating strains. A DNA-guided CRISPR system can be quickly updated to target new mutations by simply changing the DNA guide sequence.
Because this system targets RNA rather than DNA, it does not create permanent mutations in the host’s genome. This “transient editing” means the treatment can be administered to neutralize a virus, and once the guide is gone, the cell returns to its original state. This significantly lowers the risk of long-term side effects or unintended genetic mutations that have plagued earlier iterations of gene therapy.
The research suggests that this approach could be used to develop a broad spectrum of “programmable” antivirals. Instead of developing a new drug for every new virus, scientists could potentially use a single CRISPR protein and simply swap out the DNA guide to match the current threat.
What remains unknown
While the laboratory results are promising, several hurdles remain before this technology reaches clinical use. The primary challenge is delivery: getting the CRISPR protein and its DNA guide into the specific cells of a living human patient without triggering an immune response. Current delivery methods, such as lipid nanoparticles, are improving, but they are not yet perfected for all organ systems.

the long-term efficacy of DNA-guided RNA targeting in human subjects has not yet been established. Researchers must determine how long the DNA guides remain active in the body and whether the immune system will eventually recognize and neutralize the CRISPR proteins themselves.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Genetic engineering and CRISPR-based therapies are currently subject to strict regulatory oversight and are primarily in research or clinical trial phases.
The next phase of development will likely involve expanding the library of targetable RNA sequences and conducting in vivo studies to test the delivery efficiency of the DNA-guided system. Official updates on clinical trials or regulatory filings regarding this specific HKUST methodology are expected as the research moves from proof-of-concept to preclinical validation.
We want to hear from you. Do you think RNA-based editing is a safer alternative to permanent DNA modification? Share your thoughts in the comments or share this story on social media.