The path from a laboratory breakthrough to a bedside treatment is rarely a straight line, particularly for small-cap biotechnology firms navigating the volatile intersection of cutting-edge science and capital markets. Inmune Bio is currently walking this tightrope with CORDStrom, its lead candidate designed to tackle some of the most stubborn neurodegenerative challenges in modern medicine.
While the company continues to push the clinical boundaries of mesenchymal stem cell (MSC) therapy, it is doing so against a backdrop of tightening funding environments that have left many biotech innovators scrambling for liquidity. To secure the future of its pipeline, Inmune Bio recently moved to fortify its supply chain, amending a critical material transfer agreement with the Anthony Nolan U.K. Cord Blood Bank. This move ensures long-term access to the high-quality cord tissue essential for the production of CORDStrom, effectively insulating the company’s technical progress from potential raw-material shortages.
For patients and clinicians, the stakes extend far beyond corporate balance sheets. The effort to harness the immunomodulatory properties of umbilical cord-derived stem cells represents a pivot in how we approach brain health—shifting from merely slowing the progression of decay to attempting to modify the environment of the central nervous system (CNS) to support survival, and repair.
The Science of CORDStrom: Beyond Simple Regeneration
As a physician, I find the mechanism behind CORDStrom particularly compelling because it moves away from the outdated notion that stem cells must simply “replace” dead neurons. In the context of neurodegeneration, the primary hurdle is often a hostile, inflammatory environment—a “cytokine storm” of sorts—that prevents the brain from healing itself.
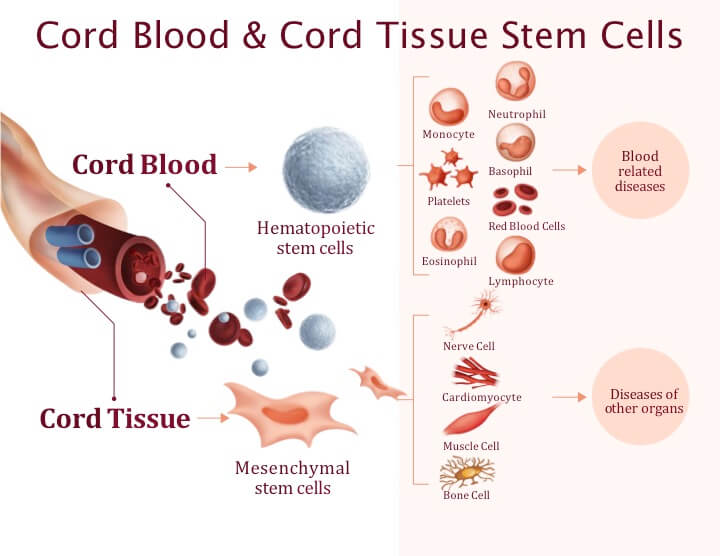
CORDStrom utilizes mesenchymal stem cells derived from the umbilical cord. Unlike hematopoietic stem cells, which form blood, MSCs act more like the body’s “pharmacies.” They secrete a potent mix of growth factors and anti-inflammatory molecules through paracrine signaling. When introduced into the system, these cells are thought to modulate the immune response, reducing the chronic inflammation that characterizes diseases like Alzheimer’s and other forms of dementia.
The decision to use cord-derived tissue over bone marrow or adipose (fat) tissue is strategic. Cord tissue is generally less immunogenic and possesses a higher proliferative capacity, meaning the cells can be expanded in a lab more efficiently while maintaining their therapeutic potency. This scalability is exactly why the agreement with the Anthony Nolan U.K. Cord Blood Bank is a cornerstone of Inmune Bio’s operational strategy.
Securing the Pipeline via Anthony Nolan
In the biotech world, a “material transfer agreement” (MTA) might sound like a mundane legal formality, but it is effectively a lifeline. For a company like Inmune Bio, the consistency of the starting material is paramount. If the source tissue varies too much in quality or biological profile, the resulting therapy can be inconsistent, leading to failed clinical trials or regulatory rejection by the FDA or EMA.
By locking in long-term access to the Anthony Nolan repository, Inmune Bio is addressing two critical risks: supply chain stability and standardization. Anthony Nolan is one of the world’s most respected registries and banks; their rigorous screening and storage protocols provide a level of biological “cleanliness” and traceability that is required for pharmaceutical-grade manufacturing.
This partnership allows Inmune Bio to focus its limited resources on clinical execution rather than the logistical nightmare of sourcing rare, high-quality biological materials on an ad-hoc basis. It transforms a variable cost and risk into a predictable asset.
The Funding Crunch and the Biotech ‘Winter’
Despite the scientific momentum, Inmune Bio is operating during what many analysts call a “biotech winter.” For the last decade, low interest rates fueled a surge of investment into speculative early-stage therapies. However, as the cost of capital has risen, investors have shifted their appetite toward companies with proven revenues or those nearing the final stages of Phase 3 trials.
Small-cap firms are now facing a “valuation gap,” where the cost of continuing research exceeds the current market valuation of the company. This puts immense pressure on management to achieve “value-inflection points”—specific clinical milestones that prove the drug works—before the cash runway expires.
The funding pressure creates a paradoxical situation: to get the funding they need, companies must provide data from large trials, but to run those trials, they need the funding. This is why strategic partnerships and efficiency-driving agreements, such as the one with Anthony Nolan, are vital. They signal to investors that the company is thinking about commercial scalability and risk mitigation, even while the treasury is lean.
| Focus Area | Current Status/Action | Strategic Importance |
|---|---|---|
| Source Material | Amended MTA with Anthony Nolan | Ensures biological consistency and scale |
| Mechanism | MSC Paracrine Signaling | Targets neuro-inflammation over cell replacement |
| Target Indication | Neurodegenerative Disorders | Addresses high unmet medical need |
| Financial State | Navigating funding pressures | Requires value-inflection milestones for capital |
What Remains Unknown
While the supply chain is secure, several critical questions remain for Inmune Bio. First is the efficacy threshold: will CORDStrom show a statistically significant improvement in cognitive function or a slowing of decline compared to placebos in a diverse patient population? Second is the regulatory pathway: the FDA has historically been cautious with stem cell therapies, requiring rigorous proof of purity and potency.
the company must demonstrate that its delivery method—how the cells reach the target areas of the brain—is both safe and effective. The blood-brain barrier remains the most formidable obstacle in neurology, and the success of CORDStrom depends heavily on the cells’ ability to exert their influence across or through this barrier.
Disclaimer: This article is for informational purposes only and does not constitute medical advice, diagnosis, or financial investment recommendation. Always seek the advice of your physician or a qualified financial advisor regarding medical or investment decisions.
The next critical checkpoint for Inmune Bio will be the release of updated clinical data or a formal filing regarding the progress of their current trial phases. These updates will likely dictate the company’s ability to secure the next round of funding or attract a larger pharmaceutical partner for co-development.
Do you believe stem cell therapy is the future of dementia care, or is the regulatory hurdle too high? Share your thoughts in the comments below.
