For many patients, surviving a myocardial infarction—a heart attack—is only the first hurdle. The real challenge often begins in the weeks that follow, as the heart attempts to heal. In some cases, the heart undergoes a process called “adverse remodeling,” where the left ventricle stretches and weakens, leading to a decline in the heart’s ability to pump blood. This transition from a heart attack to chronic heart failure is a leading cause of long-term disability and death.
A new therapeutic approach is attempting to interrupt this process by targeting the very blueprints of cellular response. The HF-REVERT trial, a randomized phase 2 study, investigated the use of CDR132L, a microRNA inhibitor designed to prevent the heart from remodeling after a major cardiac event. By targeting a specific molecule called miR-132, researchers hope to stabilize the heart’s structure and preserve its function during the critical window of recovery.
As a physician, I have seen how devastating the “post-MI dip” in ejection fraction can be for a patient’s quality of life. While contemporary standard-of-care treatments—such as beta-blockers and ACE inhibitors—are highly effective, they often manage the symptoms of remodeling rather than stopping the molecular trigger. The HF-REVERT study represents a shift toward precision medicine, attempting to “silence” the genetic signals that tell the heart to enlarge and weaken.
The Molecular Trigger: What is miR-132?
To understand CDR132L, one must first understand microRNAs. These are small, non-coding RNA molecules that act as “dimmer switches” for genes. In the wake of a heart attack, the heart produces an excess of miR-132. While this might be part of an initial stress response, prolonged elevation of miR-132 is linked to the degradation of the heart’s structural integrity and the progression toward heart failure.
CDR132L is an antisense oligonucleotide—essentially a mirror image of the target RNA—that binds to miR-132 and neutralizes it. The goal is to prevent the heart from expanding too much (dilation) and to maintain the left ventricular ejection fraction (LVEF), which is the percentage of blood the heart pumps out with each beat. When LVEF drops below 45%, the risk of heart failure increases significantly.
Inside the HF-REVERT Trial Design
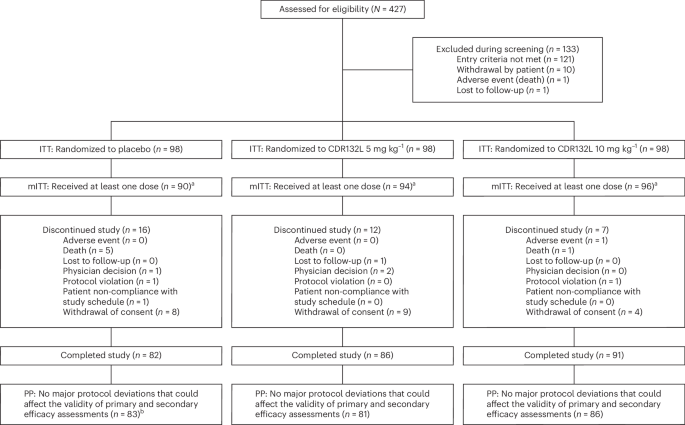
The HF-REVERT trial was designed as a rigorous, international effort to determine if CDR132L could meaningfully alter the trajectory of heart recovery. Conducted across 54 study sites in eight countries—including the United Kingdom, Germany, Spain, and the Netherlands—the trial focused on a high-risk population: adults aged 30 to 80 who had suffered a spontaneous type 1 myocardial infarction and showed early signs of systolic dysfunction.
The study employed a double-blind, placebo-controlled structure to ensure the highest level of objectivity. Patients were randomized into one of three groups: a placebo group, a low-dose group (5 mg/kg), or a high-dose group (10 mg/kg). The treatment consisted of three intravenous infusions administered 28 days apart, layered on top of the standard medical therapy patients were already receiving.
To eliminate bias, the trial utilized a blinded core laboratory at Brigham and Women’s Hospital and Harvard Medical School. This ensured that the echocardiograms used to measure the heart’s volume and function were interpreted by experts who did not know which treatment the patient had received.
| Trial Parameter | Specification |
|---|---|
| Primary Endpoint | % change in LVESVI from baseline to month 6 |
| Patient Criteria | LVEF ≤ 45% and NT-proBNP ≥ 125 pg/ml |
| Dosing Regimen | 3 IV infusions, 28 days apart |
| Study Duration | 6-month treatment + 6-month follow-up |
| Geographic Scope | 8 countries (EU and UK) |
Measuring Success: Beyond the Ejection Fraction
While the general public often focuses on “ejection fraction,” the HF-REVERT trial looked deeper at a metric called LVESVI (Left Ventricular End-Systolic Volume Index). LVESVI measures the volume of the left ventricle at the end of a contraction. If this volume increases, It’s a clear sign that the heart is dilating—stretching out like an overused balloon. Reducing this expansion is the primary goal of the therapy.
The study also monitored several secondary biomarkers and clinical outcomes to create a full picture of patient health:
- NT-proBNP: A protein released when the heart is under stretch or stress; lower levels generally indicate better heart function.
- Global Longitudinal Strain (GLS): A more sensitive measure of how the heart muscle actually contracts.
- KCCQ Score: A patient-reported questionnaire measuring the impact of heart failure on daily life and symptoms.
- Troponin T: A marker used to track ongoing myocardial injury.
By combining these hard data points with patient-reported quality of life, the trial sought to determine if the molecular inhibition of miR-132 translated into a tangible benefit for the person living with the condition.
Constraints and Considerations
Despite the promise of RNA-based therapies, the HF-REVERT trial faced specific constraints. The study excluded patients with severe renal impairment, unstable psychiatric conditions, or those who had already experienced a severe drop in ejection fraction (below 30%) within six months. This means the results primarily apply to those in the early stages of post-MI dysfunction—the “golden window” where intervention is most likely to prevent permanent failure.
the study noted a limitation regarding gender data. While exploratory analyses were performed, the small number of women enrolled in the trial means that the findings may not be fully generalizable across all sexes. This remains a common challenge in cardiovascular research, where women are often underrepresented in early-phase trials.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Patients should consult with their cardiologist or primary care physician regarding treatment options after a myocardial infarction.
The next critical step for CDR132L will be the analysis of the long-term 12-month follow-up data and the potential transition into a larger Phase 3 trial. These future studies will be essential to determine if the reduction in heart remodeling leads to a decrease in hospitalizations and overall mortality.
Do you think precision genetic medicine is the future of cardiology? Share your thoughts in the comments below.