For decades, the conversation around obesity was dominated by a frustratingly simple narrative: eat less, move more. For millions of people, this “willpower” model failed not because of a lack of effort, but because of a fundamental misunderstanding of human biology. The arrival of GLP-1 receptor agonists—marketed under household names like Ozempic, Wegovy, and Zepbound—has fundamentally shifted that paradigm, moving the needle from moral failing to metabolic management.
These medications are not merely “weight loss drugs”; they are sophisticated hormonal tools that recalibrate the body’s relationship with hunger and insulin. However, as these prescriptions move from specialized clinics into the mainstream, a critical gap has emerged between rapid weight loss and sustainable health. The goal, as clinicians are increasingly emphasizing, is not just a lower number on the scale, but the preservation of lean muscle mass and the improvement of overall metabolic function.
As a physician, I have watched the discourse around these drugs fluctuate between breathless miracle-cure narratives and skeptical dismissals. The reality lies in the nuance. While the efficacy of semaglutide and tirzepatide in reducing body weight is clinically undeniable, the quality of that weight loss determines whether the patient achieves long-term vitality or simply a smaller version of their former self, potentially with a higher percentage of body fat and lower physical strength.
The Biology of Satiety: How GLP-1s Actually Work
To understand why these drugs are so effective, one must understand the Glucagon-like peptide-1 (GLP-1) hormone. Naturally produced in the gut, GLP-1 is released after eating to signal the pancreas to release insulin and the brain to feel full. It also slows “gastric emptying,” meaning food stays in the stomach longer, extending the feeling of satiety.
Drugs like Ozempic and Wegovy (semaglutide) mimic this hormone but are engineered to last much longer in the body than the natural version. Tirzepatide (Mounjaro and Zepbound) takes this a step further by acting as a dual agonist, mimicking both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide). This dual action often results in even more significant weight loss and improved glucose control by targeting multiple metabolic pathways simultaneously.
The result is a silencing of “food noise”—that persistent, intrusive craving for high-calorie foods that often overrides rational decision-making in people with obesity. By lowering the biological drive to overeat, these medications allow patients to adhere to caloric deficits that previously felt unsustainable.
Beyond the Scale: Cardiovascular and Metabolic Gains
While the public focus remains on aesthetics and weight loss, the most significant clinical victories are happening under the skin. Obesity is a systemic inflammatory disease that puts immense strain on the heart, kidneys, and joints. The reduction in weight provided by GLP-1s triggers a cascade of systemic improvements.
One of the most pivotal findings in recent years came from the SELECT clinical trial, which demonstrated that semaglutide (Wegovy) significantly reduced the risk of major adverse cardiovascular events—such as heart attack and stroke—in adults with overweight or obesity and established cardiovascular disease, regardless of how much weight they actually lost. This suggests that GLP-1s provide cardioprotective benefits that may be independent of weight loss, potentially by reducing systemic inflammation and improving blood pressure.
the impact on Type 2 diabetes management has been transformative. By improving insulin sensitivity and reducing hepatic glucose production, these drugs help patients achieve HbA1c levels that were previously unreachable for some, reducing the long-term risk of neuropathy, retinopathy, and kidney failure.
The ‘Muscle Gap’ and the Risk of Sarcopenia
Despite the benefits, there is a hidden cost to rapid weight loss: the loss of lean muscle mass. When the body drops weight quickly, it doesn’t just burn fat; it breaks down muscle tissue for energy. This phenomenon, known as sarcopenia, can lead to a drop in basal metabolic rate (BMR), making it significantly easier to regain weight once the medication is stopped.
This is where the “magic pill” narrative becomes dangerous. Without a deliberate strategy to protect muscle, patients may experience “Ozempic face”—a gaunt appearance caused by the loss of subcutaneous fat and muscle tone in the face—and a general loss of physical strength. To combat this, medical guidance has shifted toward a mandatory “protein-first” approach.
To maintain lean mass while on a GLP-1, patients are encouraged to focus on two non-negotiable pillars:
- High Protein Intake: Prioritizing lean proteins (chicken, fish, tofu, legumes) to provide the amino acids necessary for muscle repair and maintenance.
- Resistance Training: Engaging in strength training (weightlifting, resistance bands, or bodyweight exercises) to signal to the body that muscle tissue is necessary and should be preserved.
Comparing the Primary GLP-1 Options
While several medications fall under the GLP-1 umbrella, they differ in their primary indications and chemical mechanisms.
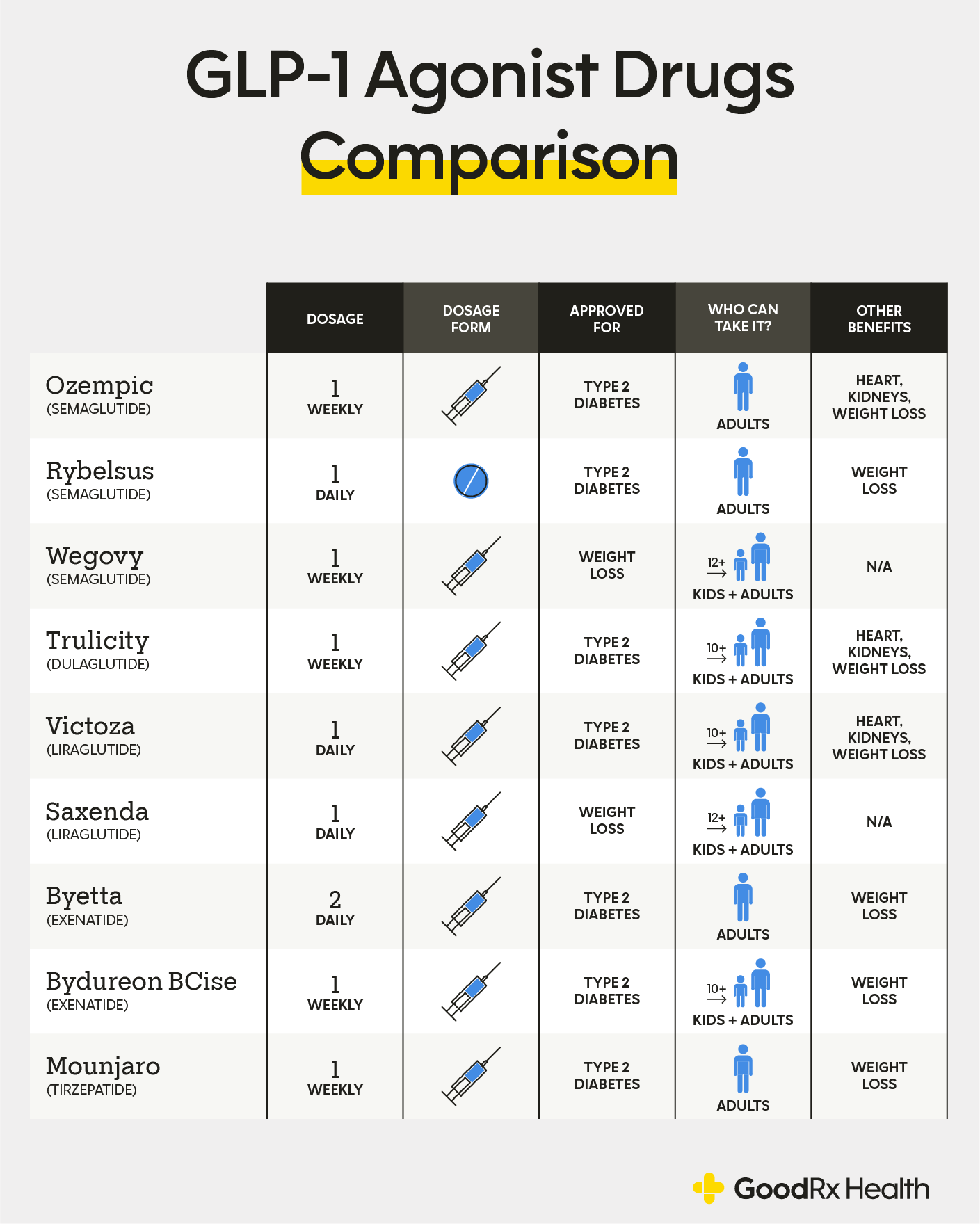
| Medication | Active Ingredient | Primary Mechanism | FDA-Approved Use |
|---|---|---|---|
| Ozempic | Semaglutide | GLP-1 Agonist | Type 2 Diabetes |
| Wegovy | Semaglutide | GLP-1 Agonist | Chronic Weight Management |
| Mounjaro | Tirzepatide | GLP-1 & GIP Agonist | Type 2 Diabetes |
| Zepbound | Tirzepatide | GLP-1 & GIP Agonist | Chronic Weight Management |
Navigating the Long-Term Path
The most pressing question facing patients today is: Is this forever? For many, obesity is a chronic condition, much like hypertension. Just as a patient would not stop taking blood pressure medication the moment their pressure normalized, stopping a GLP-1 without a rigorous maintenance plan often leads to weight regain.
The transition from the “weight loss phase” to the “maintenance phase” is the most critical window of treatment. This involves slowly tapering dosages or moving to a less frequent schedule while doubling down on the lifestyle habits—protein intake and strength training—that were established during the initial phase. The goal is to transition from a drug-dependent state to a biologically supported state where the body’s new set point is maintainable.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or treatment.
Looking ahead, the medical community is awaiting further data on “next-generation” GLP-1s, including CagriSema, which aims to combine semaglutide with a long-acting amylin analogue to further increase weight loss while potentially minimizing muscle loss. Clinical trials will continue to monitor the long-term effects of these drugs over decades, rather than years, to fully understand their impact on longevity and overall healthspan.
Do you have questions about metabolic health or your experience with GLP-1 medications? Share your thoughts in the comments below or share this article with someone navigating their health journey.