The process of building complex medicines often resembles a high-stakes puzzle where the pieces—biological proteins and chemical drugs—refuse to fit together precisely. For years, scientists have struggled with “bioconjugation,” the act of linking a therapeutic drug to a targeting molecule like an antibody. When these links are imprecise, the resulting medicine can be unstable, toxic, or simply ineffective.
A breakthrough in chemical biology has introduced a fresh molecular handle for complex medicines, utilizing a common amino acid to create a precise, predictable docking point for drug attachment. By leveraging the natural properties of proteins, researchers have developed a method to “snap” therapeutic payloads onto specific sites without compromising the protein’s structural integrity or biological function.
This advancement addresses a critical bottleneck in the production of antibody-drug conjugates (ADCs), a class of “guided missile” drugs designed to kill cancer cells although leaving healthy tissue untouched. Traditionally, attaching drugs to antibodies resulted in a chaotic mix of molecules with varying amounts of drug attached—a problem known as a fluctuating drug-to-antibody ratio (DAR)—which complicated regulatory approval and clinical dosing.
Solving the Precision Problem in Bioconjugation
At the heart of this innovation is the strategic use of cysteine, a common amino acid found in almost all proteins. While cysteine has long been used in laboratories for its reactive sulfur group, it is often too reactive or poorly positioned to allow for the surgical precision required in modern pharmacology.
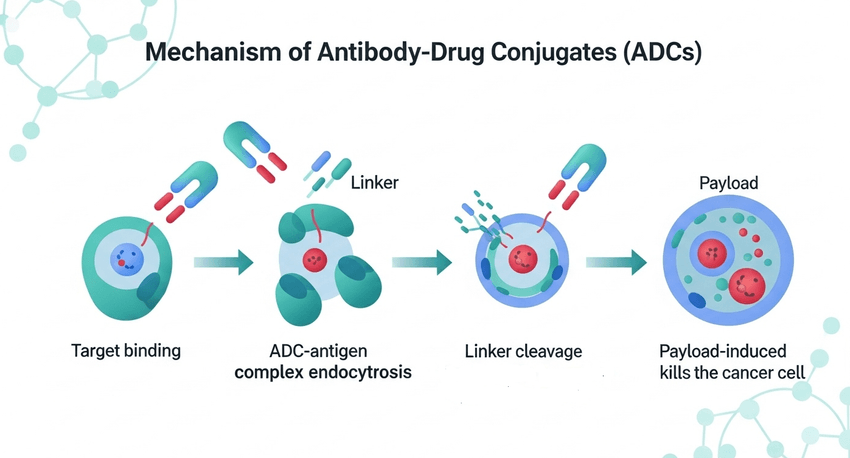
The new method transforms this amino acid into a sophisticated “handle.” By manipulating the chemical environment around the cysteine residue, researchers can ensure that the drug attaches only to the intended spot. This site-specific attachment ensures that every single molecule of the medicine is identical, a requirement for the U.S. Food and Drug Administration (FDA) and other regulators who demand strict consistency in pharmaceutical manufacturing.
From a software engineering perspective—a lens I often apply to these biological systems—this is akin to moving from a “loose” data structure to a strictly typed one. Instead of hoping the drug lands in the right place, scientists now have a defined address for the payload, reducing “noise” and increasing the efficiency of the therapeutic delivery.
Comparing Conjugation Methods
The shift from random to site-specific conjugation represents a fundamental change in how complex biologics are engineered. The following table outlines the primary differences between traditional methods and the new molecular handle approach.
| Feature | Traditional Conjugation | Molecular Handle Method |
|---|---|---|
| Attachment Site | Random/Multiple | Site-Specific/Defined |
| Product Consistency | Heterogeneous Mix | Homogeneous Population |
| Drug-to-Antibody Ratio | Variable (Stochastic) | Precise and Fixed |
| Manufacturing Complexity | High Purification Needed | Streamlined Synthesis |
Impact on Targeted Drug Delivery
The implications for oncology and autoimmune research are significant. When a drug is attached randomly to an antibody, it can block the antibody’s “business complete”—the part that recognizes and binds to the cancer cell. This renders the medicine useless or, worse, causes it to detach prematurely in the bloodstream, leading to systemic toxicity.
By using a dedicated molecular handle, the drug is tucked away in a position that does not interfere with the antibody’s targeting capabilities. This allows for a higher payload of the drug to be delivered directly into the diseased cell, potentially lowering the overall dose required for the patient and reducing side effects.
Beyond cancer, this technology opens doors for other complex medicines, including:
- Enzyme-Prodrug Conjugates: Delivering inactive drugs that are only activated by specific enzymes within a tumor.
- Imaging Agents: Attaching fluorescent dyes or radioactive tracers to proteins for high-resolution diagnostic imaging.
- Protein Scaffolds: Building multi-functional proteins that can perform several biological tasks simultaneously.
Overcoming Manufacturing Constraints
One of the most persistent challenges in biotech is scalability. Many previous attempts at site-specific conjugation required “unnatural” amino acids—synthetic building blocks that the cell does not produce naturally. Incorporating these into a protein often required complex genetic engineering or expensive chemical synthesis that failed when scaled up for mass production.
Due to the fact that this new approach relies on a common amino acid, it integrates seamlessly into existing cellular production lines. This means that pharmaceutical companies can potentially adopt the technology without rebuilding their entire manufacturing infrastructure, significantly shortening the timeline from laboratory discovery to clinical trials.
The process effectively utilizes “click chemistry” principles—a field of chemistry that allows molecules to be joined quickly and reliably, similar to how a seatbelt clicks into place. By creating a compatible “socket” using the common amino acid, the drug can be attached in a single, efficient step with nearly 100% yield.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
The next milestone for this technology will be the transition into human clinical trials to verify if the increased precision of these “handled” molecules translates to better patient outcomes compared to first-generation ADCs. Researchers are expected to publish updated stability and toxicity data as these candidates move through the pre-clinical pipeline.
Do you think site-specific precision will finally make targeted chemotherapy a standard for all cancer types? Share your thoughts in the comments or share this story with your network.

