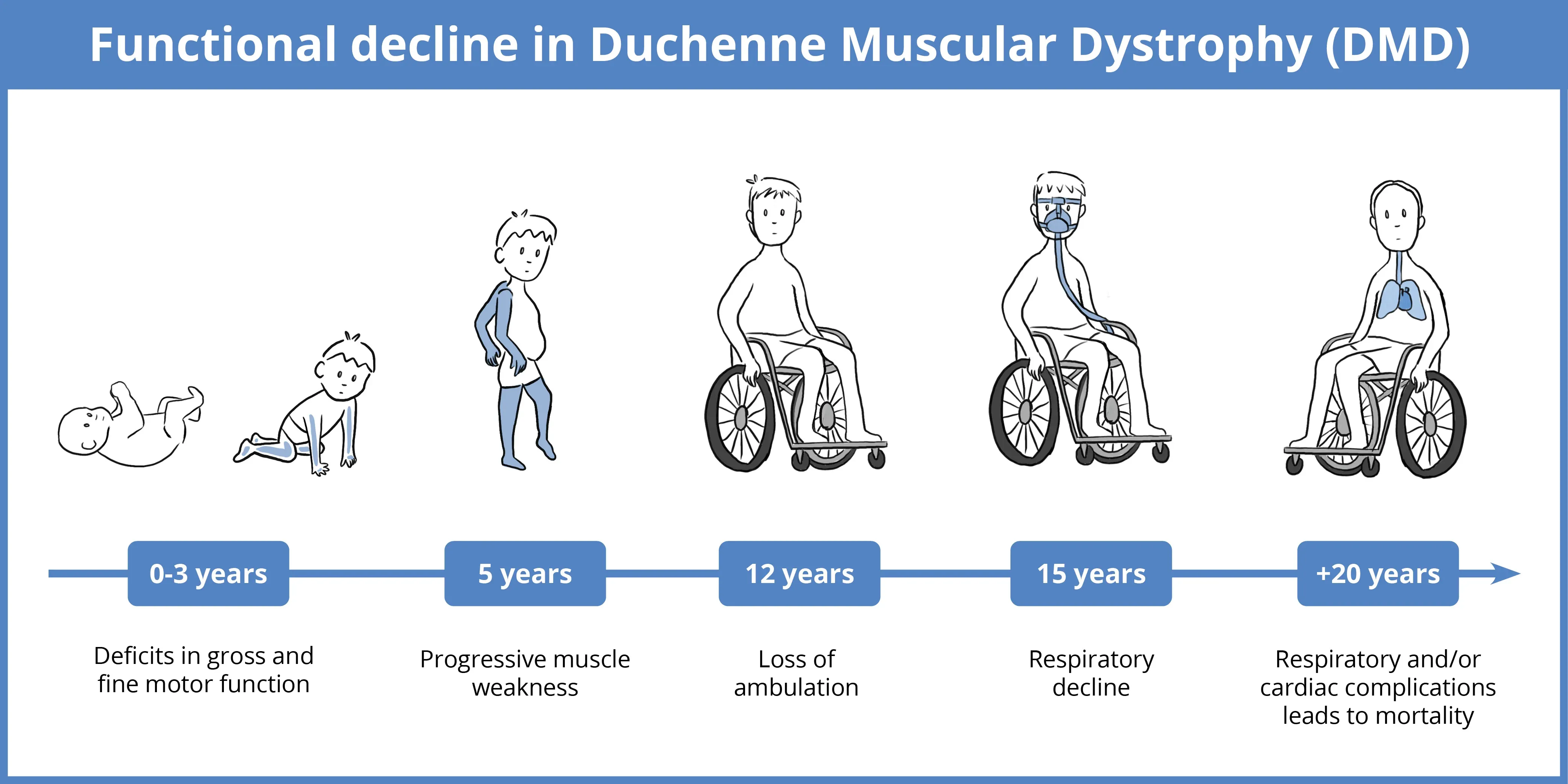
For families living with Duchenne muscular dystrophy (DMD), time is the most precious and precarious commodity. The disease, a genetic condition that causes progressive muscle degeneration, follows a heartbreakingly predictable trajectory: a childhood of fading strength, the eventual loss of the ability to walk, and a premature decline in respiratory and cardiac function.
A recent recommendation from the National Institute for Health and Care Excellence (NICE) offers a significant shift in that timeline. The agency has recommended the use of givinostat, a novel medication that could delay the loss of ambulation—the transition to a wheelchair—by up to five years for some patients. The decision opens the door for approximately 530 patients across England to access a treatment that targets the disease not by fixing the genetic mutation, but by altering the environment in which the muscles struggle to survive.
Unlike many recently publicized DMD therapies that target specific genetic mutations or attempt to replace the missing dystrophin protein, givinostat is recommended for all types of DMD. This broad eligibility is a critical victory for patient advocacy groups, as it provides a therapeutic option for children who do not qualify for mutation-specific “exon-skipping” drugs or certain gene therapies.
Changing the Muscle Environment: How Givinostat Works
As a physician, I find the mechanism of givinostat particularly compelling because it addresses the “collateral damage” of DMD. In a healthy body, the protein dystrophin acts as a shock absorber for muscle fibers. Without it, muscles suffer repeated micro-tears, leading to chronic inflammation and the eventual replacement of muscle tissue with fat and scar tissue (fibrosis).
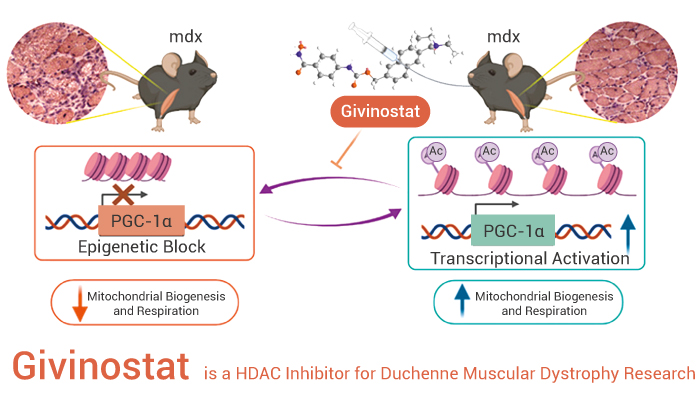
Givinostat is a histone deacetylase (HDAC) inhibitor. Rather than attempting to rewrite the genetic code, it works at the epigenetic level to modulate the expression of genes involved in inflammation, and fibrosis. By inhibiting HDAC, the drug helps reduce the inflammatory response and may promote the survival of existing muscle fibers. It doesn’t cure the disease, but it slows the rate at which the body destroys its own muscle tissue.
This approach complements the current gold standard of care—corticosteroids. While steroids are effective at maintaining strength, they carry a heavy burden of side effects, including stunted growth, weight gain, and bone density loss. Givinostat provides a different pharmacological pathway to preserve function, potentially allowing patients to remain mobile longer into their adolescence.
Analyzing the PROMOSE Evidence
The NICE recommendation is grounded in data from the PROMOSE trial, a pivotal study that compared givinostat to a placebo in boys with DMD. The trial focused on the “time to loss of ambulation,” a primary endpoint that serves as a proxy for quality of life and independence.
The data indicated that patients receiving givinostat maintained their ability to walk significantly longer than those in the control group. The “five-year delay” cited in clinical assessments represents the potential window of extended mobility. For a teenager, five additional years of walking can mean the difference between attending a mainstream school independently or requiring full-time caregiver support.
However, the path to approval was not without scrutiny. NICE evaluators had to weigh the clinical benefit against the cost-effectiveness of the drug. The agency typically requires a “cost per quality-adjusted life year” (QALY) that falls within a specific threshold. To meet these requirements, the recommendation is often contingent on a commercial agreement—essentially a discount provided by the manufacturer, Italfarmaco, to ensure the NHS can provide the drug sustainably.
| Treatment Type | Primary Goal | Eligibility | Key Limitation |
|---|---|---|---|
| Corticosteroids | Reduce inflammation | Broad/General | Significant systemic side effects |
| Exon-Skipping | Restore partial protein | Mutation-specific | Limited to specific genetic subsets |
| Givinostat | Epigenetic modulation | All DMD types | Delays, rather than stops, progression |
| Gene Therapy | Protein replacement | Specific mutations/age | High cost; long-term durability unknown |
Impact on Patients and the Healthcare System
The immediate impact of this decision is the creation of a new standard of care for roughly 530 children and adolescents in England. For these patients, the recommendation removes the bureaucratic and financial hurdles that previously made such an innovative therapy inaccessible.
The stakeholders in this decision include not only the patients and their families but also the multidisciplinary teams—neurologists, physiotherapists, and cardiologists—who manage DMD. By delaying the loss of ambulation, the healthcare system may see a delayed need for expensive mobility equipment and a potential shift in the timing of respiratory interventions.
Despite the optimism, some unknowns remain. While the PROMOSE trial provided strong evidence for mobility, the long-term effects of HDAC inhibition over decades are still being studied. Clinicians will be monitoring whether the delay in walking correlates with a similar delay in the decline of cardiac and pulmonary function, which are the ultimate determinants of life expectancy in DMD.
For those seeking official guidance or eligibility criteria, the full NICE technology appraisal can be accessed via the NICE website, where the specific parameters for prescribing are detailed for NHS trusts.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Patients and caregivers should consult with a qualified healthcare provider to determine the most appropriate treatment plan for their specific medical condition.
The next critical step is the implementation phase, as NHS trusts begin integrating givinostat into their prescribing protocols. Official updates on the rollout and patient access timelines are expected as the commercial agreements between the NHS and Italfarmaco are finalized.
Do you have questions about the latest developments in neuromuscular therapies? Share this article or leave a comment below to join the conversation.
