For many cancer survivors, the end of primary treatment marks the beginning of a new chapter in health. However, for some patients treated with radiation for sarcoma, the long-term effects of therapy can create silent vulnerabilities in the skeletal system, leading to severe complications years after the initial diagnosis.
A recent clinical case underscores the complex relationship between radiation therapy and bone health, detailing a patient who developed chronic osteomyelitis following radiation-treated sarcoma. The case highlights a dangerous progression where radiation-induced damage predisposed the bone to a deep-seated infection, eventually resulting in a pathologic fracture—a break that occurs in bone already weakened by disease.
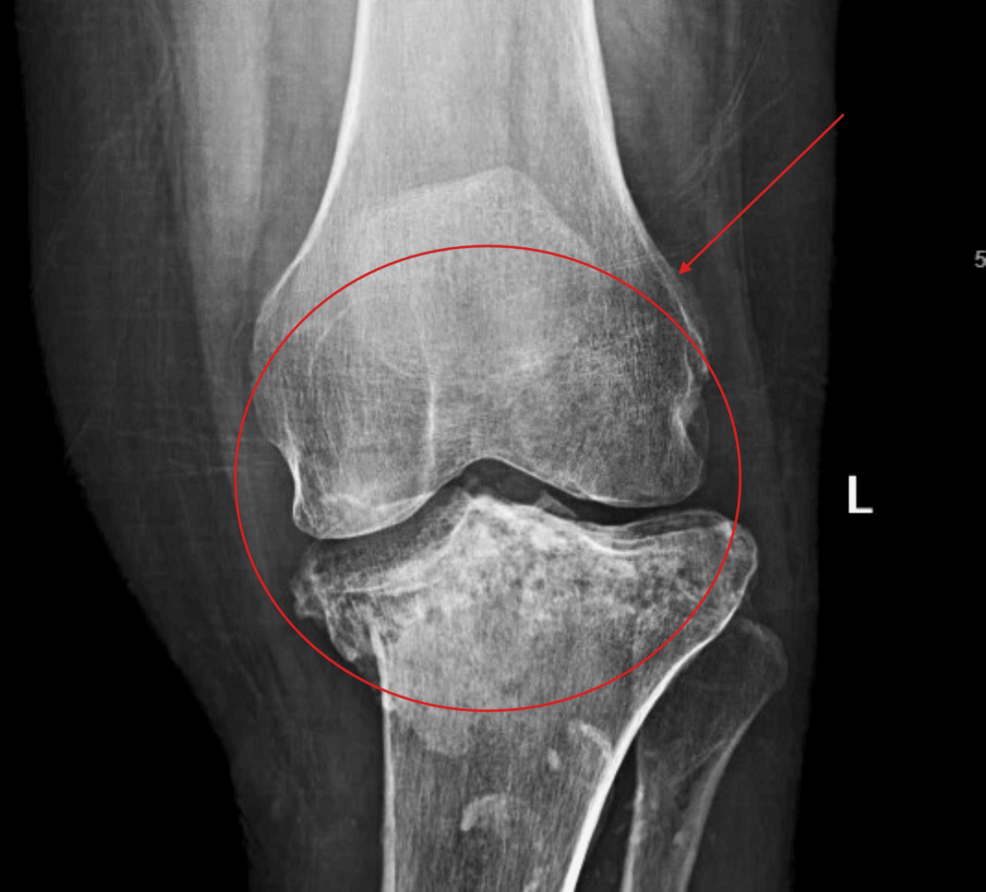
The case involved a 54-year-old male with a history of sarcoma who had previously undergone radiation therapy. He presented with knee effusion—an abnormal accumulation of fluid in the joint—which served as an early warning sign of a deeper, more systemic failure of the bone tissue. What began as joint swelling evolved into a chronic bone infection and a structural collapse of the limb, necessitating an aggressive multidisciplinary surgical intervention.
The Sentinel Symptom: Understanding Knee Effusion
In the context of post-radiation recovery, a swollen joint is rarely just a sign of wear and tear. In this patient’s case, the knee effusion was the primary clinical indicator that something was wrong beneath the surface. When fluid builds up in the joint space, it often signals inflammation, injury, or infection.
For patients with a history of radiation, this inflammation can be a harbinger of osteoradionecrosis—a condition where bone tissue dies due to radiation-induced damage to the blood supply. This necrotic tissue becomes a breeding ground for bacteria, as the compromised blood flow prevents the body’s immune cells and systemic antibiotics from reaching the site of infection effectively.
The progression from effusion to a pathologic fracture is a critical window for intervention. A pathologic fracture differs from a standard break due to the fact that it requires very little trauma to occur. the bone is so structurally compromised by the underlying osteomyelitis and radiation damage that it can fail under the stress of normal daily activities.
The Pathophysiology of Radiation-Induced Bone Failure
To understand why radiation therapy can lead to chronic osteomyelitis, it is necessary to look at the cellular impact of ionizing radiation on bone marrow and vasculature. Radiation therapy is essential for shrinking tumors and preventing sarcoma recurrence, but it can cause permanent changes to the surrounding healthy tissue.
The process typically follows a specific biological sequence:
- Endarteritis Obliterans: Radiation causes inflammation and narrowing of the small arteries within the bone.
- Hypoxia: As blood vessels close, the bone tissue is deprived of oxygen and essential nutrients.
- Tissue Necrosis: The lack of oxygen leads to the death of osteocytes (bone cells), creating areas of dead bone known as sequestra.
- Infection Susceptibility: These dead zones lack a blood supply, making them “immunologically silent” areas where bacteria can proliferate without interference from the host’s immune system.
This environment allows for the development of chronic osteomyelitis, a persistent infection that is notoriously difficult to treat because antibiotics cannot easily penetrate the necrotic, avascular bone.
Timeline of Clinical Progression
| Stage | Clinical Presentation | Underlying Pathology |
|---|---|---|
| Initial Phase | Post-Radiation Recovery | Vascular damage and latent bone hypoxia |
| Early Warning | Knee Effusion & Pain | Inflammatory response to bone necrosis/infection |
| Chronic Phase | Persistent Infection | Establishment of chronic osteomyelitis |
| Critical Event | Pathologic Fracture | Structural failure of radiation-weakened bone |
Surgical Intervention and Long-Term Management
Treating chronic osteomyelitis in a radiation-damaged limb requires more than just medication. Because the blood supply is compromised, systemic antibiotics often fail to clear the infection entirely. The gold standard for treatment involves a combination of surgical debridement and mechanical stabilization.
Surgical debridement involves the physical removal of all necrotic bone and infected tissue. By clearing out the dead “sequestra,” surgeons can create a healthier environment that allows remaining blood vessels to potentially reorganize and allows antibiotics to reach the site of infection. In this case, the presence of a pathologic fracture added a layer of complexity, requiring the surgeons to stabilize the bone—often using internal fixation (plates and screws) or external fixators—to restore the limb’s structural integrity.
The recovery process for these patients is typically prolonged. It often involves long-term intravenous antibiotic therapy and rigorous physical therapy to regain joint mobility and strength in the affected limb.
Implications for Sarcoma Survivors
This case serves as a critical reminder for the oncology and orthopedic communities regarding the long-term surveillance of sarcoma patients. The window for successful intervention is narrow; once a pathologic fracture occurs, the morbidity increases significantly, and the path to recovery becomes more arduous.
Medical providers are encouraged to maintain a high index of suspicion when post-radiation patients present with non-specific joint pain or effusion. Rather than attributing these symptoms to general arthritis or age, clinicians must consider the possibility of radiation-induced necrosis and secondary infection.
For patients, the key takeaways include:
- Monitoring: Reporting any new swelling, warmth, or persistent pain in areas that received radiation.
- Vigilance: Understanding that complications can arise years after the completion of cancer treatment.
- Integrated Care: Ensuring that orthopedic surgeons and oncologists are communicating throughout the survivorship phase.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Patients should consult with a licensed healthcare provider for diagnosis and treatment of any medical condition.
The next phase of clinical focus for these types of complications involves the development of more targeted delivery systems for antibiotics, such as antibiotic-loaded bone cement or biodegradable beads, which can provide high concentrations of medication directly to the infected site without relying on a compromised blood supply. Ongoing research into regenerative medicine and bone grafting also aims to restore the vascularity of radiation-damaged limbs.
Do you have experience navigating long-term recovery after cancer treatment? Share your thoughts or questions in the comments below.