A Houston physician who became a flashpoint of medical controversy during the COVID-19 pandemic is once again stepping into the spotlight, this time targeting a rare but deadly viral threat. Dr. Mary Talley Bowden is now offering to sell ivermectin directly to Texas residents, coinciding with a surge in public concern over hantavirus following a recent cruise ship outbreak.
The move marks a bold escalation for Bowden, who has long challenged federal health guidelines. By offering the drug for sale without a prescription, she is leveraging a shift in Texas state law to bypass traditional pharmaceutical channels, while simultaneously claiming the antiparasitic medication could be an effective shield against hantavirus.
As a board-certified physician, I have watched the ivermectin debate evolve from a clinical question into a political symbol. However, the transition from treating parasitic worms to treating acute viral syndromes is a leap that lacks the support of rigorous, peer-reviewed clinical evidence. While the drug is a cornerstone of antiparasitic medicine, its application as a broad-spectrum antiviral in humans remains unproven and unauthorized by the FDA.
A New Target for an Old Controversy
The current friction began in early May 2026, when Bowden took to X (formerly Twitter) to suggest that ivermectin “should work” against hantavirus. Her claims came just as health officials were monitoring Texas residents linked to a hantavirus outbreak on a cruise ship. Although the Texas Department of State Health Services confirmed that the affected residents were asymptomatic, the news sparked a wave of anxiety among the public.
Bowden’s rationale rests on the biological classification of the virus. In her posts, she argues that because hantavirus is an RNA virus, ivermectin can disrupt its life cycle.
Shortly after these claims, Bowden announced she would sell the drug directly to Texans, offering 100-count bottles of 12 mg and 18 mg tablets for $85 and $110, respectively. She emphasized that no prescription would be required for these sales, explicitly limiting her customer base to residents of Texas.
The Medical Reality: Hantavirus vs. Ivermectin
To understand why public health experts are pushing back, it is necessary to look at what hantavirus actually is. Hantavirus Pulmonary Syndrome (HPS) is a severe respiratory disease typically transmitted to humans through contact with the urine, droppings, or saliva of infected rodents. It is characterized by a rapid progression from flu-like symptoms to severe shortness of breath and respiratory failure.
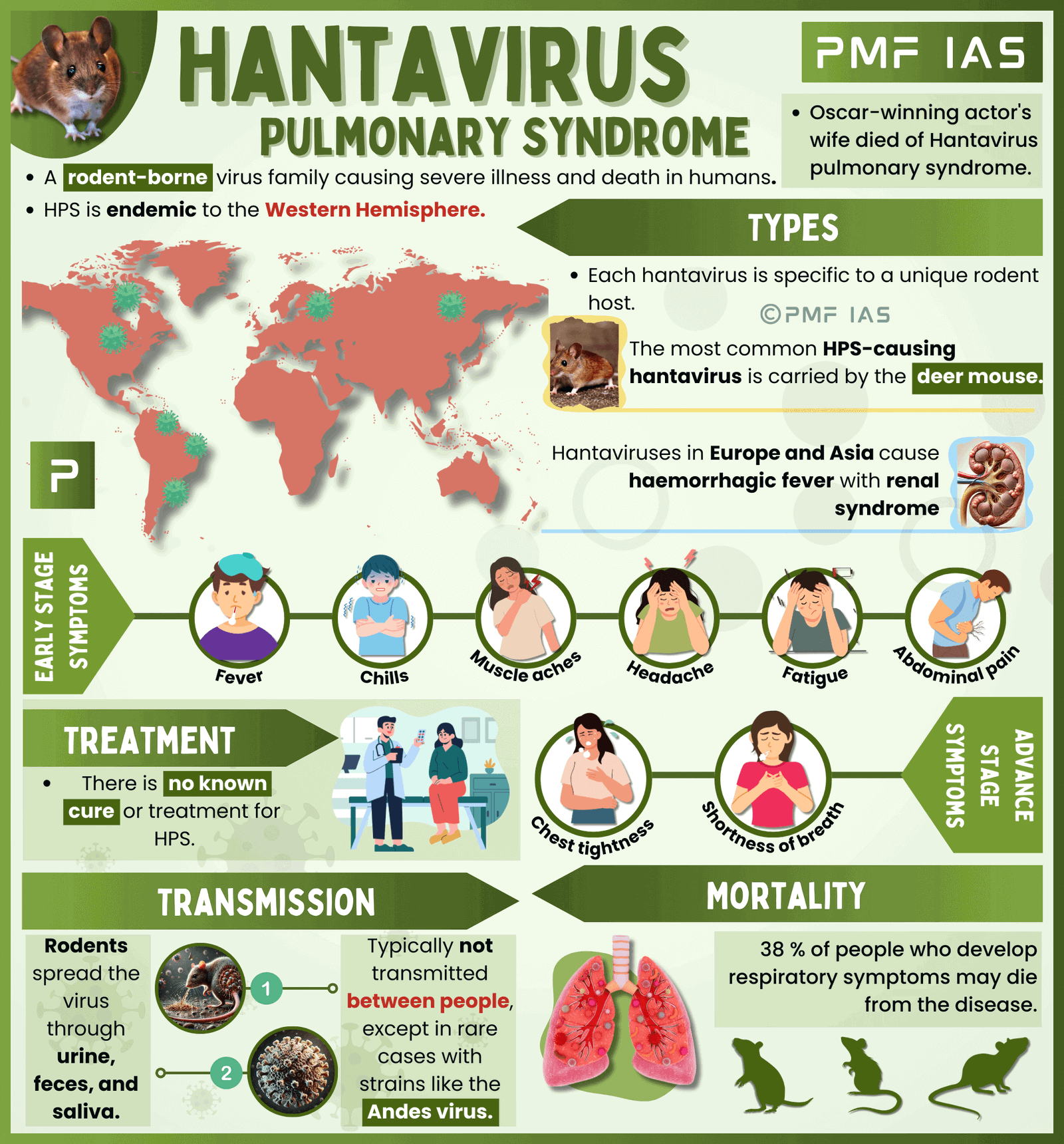
While Bowden points to the “RNA virus” nature of hantavirus as a justification for using ivermectin, this is a significant oversimplification. Many different types of viruses share an RNA structure, but they interact with human cells in vastly different ways. There is currently no clinical data demonstrating that ivermectin can prevent or treat hantavirus in humans.
The FDA’s approved uses for ivermectin are specific and well-documented. The following table outlines the gap between regulatory approval and the claims currently being promoted:
| Application | FDA Approval Status | Clinical Basis |
|---|---|---|
| Parasitic Worms | Approved (Oral) | Strong evidence for antiparasitic efficacy. |
| Head Lice / Rosacea | Approved (Topical) | Effective for localized skin/scalp treatment. |
| COVID-19 | Not Authorized | Lack of evidence in large-scale randomized trials. |
| Hantavirus | Not Approved | No established clinical evidence for efficacy. |
A Legal Battleground in the Lone Star State
Bowden’s ability to sell ivermectin without a prescription is not an accident; it is the result of a shifting legal landscape in Texas. In September 2025, Governor Greg Abbott signed House Bill 25 into law during the state’s 89th legislative session. This legislation allows pharmacists to dispense ivermectin without a traditional prescription, a move that critics argue undermines patient safety and medical oversight.
This legislative shift has provided a shield for Bowden, who has long been at odds with the medical establishment. During the COVID-19 pandemic, she was a prominent advocate for ivermectin, which eventually led to her resigning her privileges at Houston Methodist in 2021 while under investigation. The Texas Medical Board (TMB) has continued to pursue disciplinary action against her, asserting that she attempted to treat patients at a hospital where she no longer held privileges.
However, Bowden has found a powerful ally in Texas Attorney General Ken Paxton. In February 2026, Paxton’s office stepped in to support Bowden, framing her struggle as a matter of “health freedom” and constitutional rights. Paxton claimed that Bowden was being subjected to an “illegal reprimand” and praised her as a “champion for health freedom.”
Despite the political support, the TMB remains resolute. In a recent statement, the board emphasized its commitment to ensuring that hospitalized Texans receive care from authorized physicians, vowing to “vigorously contest” any attempts to overturn their disciplinary actions.
Public Reaction and Patient Risk
The reaction to Bowden’s direct-sales announcement has been sharply divided. While some supporters have pleaded with her to expand her sales beyond Texas, others have questioned how she maintains an active medical license while promoting unapproved treatments and selling medications directly to the public.
From a public health perspective, the danger lies in the “opportunity cost” of treatment. When patients pursue unproven remedies for a disease as aggressive as hantavirus, they may delay seeking the critical supportive care—such as mechanical ventilation—that is often the only way to survive the illness. Taking medications without professional dosage oversight increases the risk of adverse reactions.
In an emailed statement, Bowden denied that her sales effort was specifically tied to the hantavirus scare, claiming she had been working to make the drug more accessible since the passage of HB 25. She argued that despite the new law, ivermectin remains “unnecessarily expensive” and tricky to obtain for many Texans.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
The conflict between the Texas Medical Board and Dr. Bowden is expected to continue as the TMB prepares for further legal filings to uphold its disciplinary standards. The next critical checkpoint will be the upcoming review of the TMB’s administrative actions, which will determine if Bowden’s promotional activities constitute a violation of professional medical ethics under state law.
We want to hear from you. Do you believe state laws should allow the sale of medications like ivermectin without a prescription? Share your thoughts in the comments below or share this story on social media to join the conversation.