A 24-year-old woman’s intentional overdose of a common mood stabilizer led to a critical medical crisis, resulting in severe hyperammonemic encephalopathy following valproate overdose. What began as a psychiatric emergency rapidly evolved into a life-threatening neurological collapse, characterized by a deep coma and dangerously high levels of ammonia in the blood.
The patient, who had been managing bipolar disorder, ingested an estimated 30 grams of sustained-release sodium valproate. While initial assessments suggested stability, her condition deteriorated sharply over the following hours. She transitioned from being conscious to a state of profound unconsciousness, with a Glasgow Coma Scale (GCS) score dropping to 3—the lowest possible rating, indicating a deep coma.
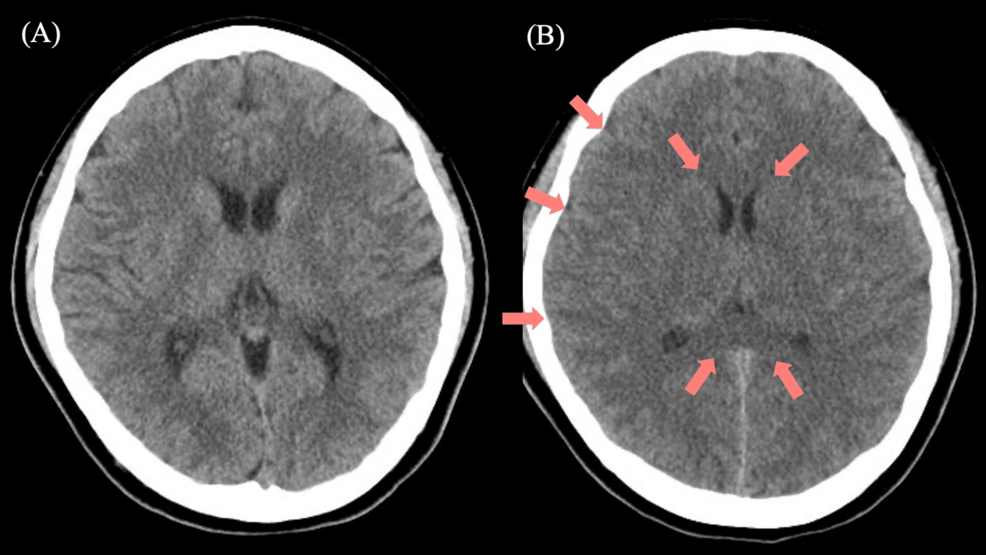
Medical teams discovered that the overdose had triggered a metabolic cascade, causing ammonia to accumulate in her system to critical levels. This condition, known as hyperammonemic encephalopathy, occurs when the liver cannot efficiently clear ammonia, allowing the toxin to cross the blood-brain barrier and cause cerebral edema, or brain swelling.
The Rapid Progression of Ammonia Toxicity
Sodium valproate is widely used to treat epilepsy and bipolar disorder, but in massive doses, it disrupts the urea cycle—the body’s primary mechanism for removing ammonia. When this cycle fails, ammonia levels spike, leading to a toxic environment for neurons. In this specific case, the patient’s ammonia levels peaked at 458 µmol/L, far exceeding the normal physiological range.
The resulting encephalopathy is not merely a sedative effect of the drug but a metabolic poisoning of the brain. The patient exhibited significant cerebral edema, which increased intracranial pressure and threatened permanent neurological damage. This progression underscores the danger of valproate toxicity, where the clinical presentation can be deceptive, appearing stable before a sudden, precipitous decline into coma.
The Complicating Factor of Sustained-Release Formulations
A critical element of this case was the apply of a sustained-release (SR) formulation of the medication. Unlike immediate-release tablets, SR versions are designed to release the drug slowly over time. In the context of an overdose, this creates a “reservoir effect,” where the drug continues to be absorbed into the bloodstream long after the initial ingestion.
This prolonged absorption window complicates emergency treatment. Standard gastric decontamination, such as activated charcoal, may be less effective if the drug has already begun its leisurely release from the tablet matrix. For clinicians, the SR formulation means the window of toxicity is extended, requiring more prolonged monitoring and a more aggressive approach to clearing the toxin from the body.
Critical Interventions: L-Carnitine and Hemodialysis
Treatment for severe valproate-induced hyperammonemia requires a multi-pronged approach to stabilize the brain and clear the metabolic waste. The medical team utilized L-carnitine therapy, a key intervention in valproate toxicity. Valproate depletes the body’s carnitine stores, which are essential for fatty acid metabolism and the clearance of valproic acid metabolites. By administering IV L-carnitine, doctors aim to restore these levels and facilitate the removal of ammonia.
However, since of the severity of the encephalopathy and the sustained-release nature of the overdose, L-carnitine alone was insufficient. The patient required urgent hemodialysis. Hemodialysis serves two primary purposes in this scenario: it directly removes the valproic acid from the blood and helps lower the systemic ammonia levels, reducing the pressure on the brain.
| Stage | Clinical Status | Primary Intervention |
|---|---|---|
| Initial Admission | Conscious, stable vitals | Observation and monitoring |
| Acute Decline | GCS 3 (Deep Coma) | Airway protection / Intubation |
| Metabolic Peak | Ammonia 458 µmol/L | IV L-carnitine administration |
| Critical Phase | Cerebral edema | Urgent hemodialysis |
| Recovery | Gradual awakening | Supportive care and psychiatric stabilization |
Clinical Implications and Patient Recovery
The patient eventually recovered, demonstrating the efficacy of aggressive metabolic intervention. This case highlights the necessity of measuring serum ammonia levels in any patient presenting with altered mental status following a valproate overdose, even if the initial drug levels do not seem immediately lethal.
The synergy between L-carnitine and hemodialysis proved decisive. While L-carnitine addresses the biochemical deficiency caused by the drug, hemodialysis provides the mechanical clearance necessary to prevent irreversible brain damage from cerebral edema. For healthcare providers, the takeaway is clear: the “sustained” nature of the medication requires a “sustained” and aggressive treatment strategy.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
Medical boards and toxicology experts continue to monitor the safety profiles of mood stabilizers, with a focus on improving rapid-response protocols for metabolic crises. The next step for clinical guidelines involves further refining the thresholds at which hemodialysis is initiated for valproate-induced hyperammonemia to ensure timely intervention across diverse patient populations.
Do you have experience with these medications or thoughts on patient safety protocols? Share your comments below or share this article with your colleagues.